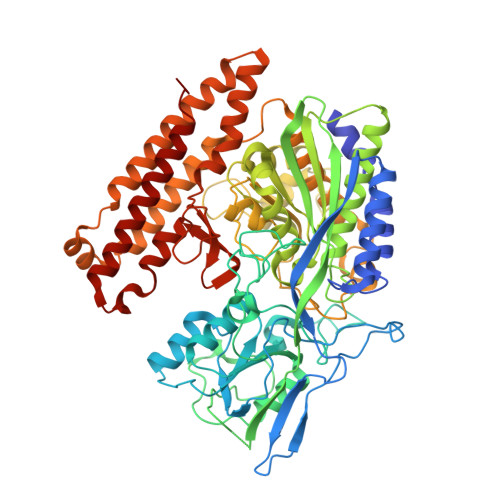
Structure of glutamate carboxypeptidase II, a drug target in neuronal damage and prostate cancer.
Mesters, J.R., Barinka, C., Li, W., Tsukamoto, T., Majer, P., Slusher, B.S., Konvalinka, J., Hilgenfeld, R.(2006) EMBO J 25: 1375-1384
- PubMed: 16467855 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/sj.emboj.7600969
- Primary Citation Related Structures:
2C6C, 2C6G, 2C6P - PubMed Abstract:
Membrane-bound glutamate carboxypeptidase II (GCPII) is a zinc metalloenzyme that catalyzes the hydrolysis of the neurotransmitter N-acetyl-L-aspartyl-L-glutamate (NAAG) to N-acetyl-L-aspartate and L-glutamate (which is itself a neurotransmitter). Potent and selective GCPII inhibitors have been shown to decrease brain glutamate and provide neuroprotection in preclinical models of stroke, amyotrophic lateral sclerosis, and neuropathic pain. Here, we report crystal structures of the extracellular part of GCPII in complex with both potent and weak inhibitors and with glutamate, the product of the enzyme's hydrolysis reaction, at 2.0, 2.4, and 2.2 A resolution, respectively. GCPII folds into three domains: protease-like, apical, and C-terminal. All three participate in substrate binding, with two of them directly involved in C-terminal glutamate recognition. One of the carbohydrate moieties of the enzyme is essential for homodimer formation of GCPII. The three-dimensional structures presented here reveal an induced-fit substrate-binding mode of this key enzyme and provide essential information for the design of GCPII inhibitors useful in the treatment of neuronal diseases and prostate cancer.
- Institute of Biochemistry, Center for Structural and Cell Biology in Medicine, University of Lübeck, Lübeck, Germany.
Organizational Affiliation: