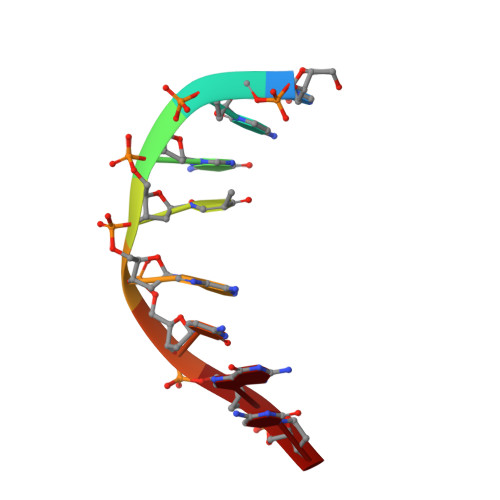
Solution structures of psoralen monoadducted and cross-linked DNA oligomers by NMR spectroscopy and restrained molecular dynamics.
Spielmann, H.P., Dwyer, T.J., Hearst, J.E., Wemmer, D.E.(1995) Biochemistry 34: 12937-12953
- PubMed: 7548052 Search on PubMed
- Primary Citation Related Structures:
203D, 204D - PubMed Abstract:
We have used two-dimensional 1H NMR spectroscopy to determine the solution structures of the 4'-(hydroxymethyl)-4,5',8-trimethylpsoralen (HMT) furanside monoadducted (MAf) and the photoisomeric HMT interstrand cross-linked (XL) DNA oligonucleotide d(5'-GCGTACGC-3')2. The determination of the structure was based on total relaxation matrix analysis of the NOESY cross-peak intensities using the program MARDIGRAS. Improved procedures to consider the experimental "noise" in NOESY spectra during these calculations have been employed. The NOE-derived distance restraints were applied in restrained molecular dynamics calculations. Twenty final structures each were generated for both the MAf and XL from both A-form and B-form dsDNA starting structures. The root-mean-square (rms) deviations of the coordinates for the 40 structures for the MAf and XL were 1.12 and 1.10 A, respectively. The rmsd of the MAf with respect to the XL is 2.20 A. The local DNA structure is distorted in both adducts, with the helix unwound by 34 degrees and 25 degrees for the MAf and XL, respectively, and an overall helical repeat of 11 base pairs, caused by intercalation of the HMT. The MAf is a photochemical intermediate on the path to interstrand XL. Considerable local structural distortion is induced by both adducts, but the DNA returns to B-form structure within three base pairs of the damage site. There is no significant bend in the helix axis of either the MAf or the XL. We have evaluated the accuracy of the two major methods of converting NOESY data into interproton distances, the isolated spin-pair approximation (ISPA) and the complete relaxation rate matrix analysis (RMA). Both methods were evaluated by comparing the resulting calculated interproton distances generated to known covalently fixed distances in the HMT. The overall structures were evaluated by checking their agreement with biophysical evidence from non-NMR techniques. Only the modified RMA method gave correct interproton distances.
- Structural Biology Division, Lawrence Berkeley Laboratory, University of California, Berkeley 94720, USA.
Organizational Affiliation: