The Structures of the Monoclinic and Orthorhombic Forms of Hen Egg-White Lysozyme at 6 Angstroms Resolution
Artymiuk, P.J., Blake, C.C.F., Rice, D.W., Wilson, K.S.(1982) Acta Crystallogr B 38: 778
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(1982) Acta Crystallogr B 38: 778
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| LYSOZYME | 129 | Gallus gallus | Mutation(s): 0 EC: 3.2.1.17 | 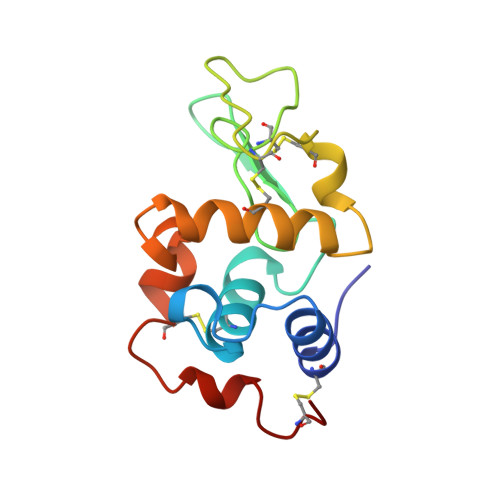 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00698 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 59.062 | α = 90 |
| b = 68.451 | β = 90 |
| c = 30.517 | γ = 90 |
| Software Name | Purpose |
|---|---|
| RIGAKU | data collection |
| bioteX | data reduction |
| X-PLOR | model building |
| GPRLSA | refinement |
| X-PLOR | refinement |
| RIGAKU | data reduction |
| bioteX | data scaling |
| X-PLOR | phasing |