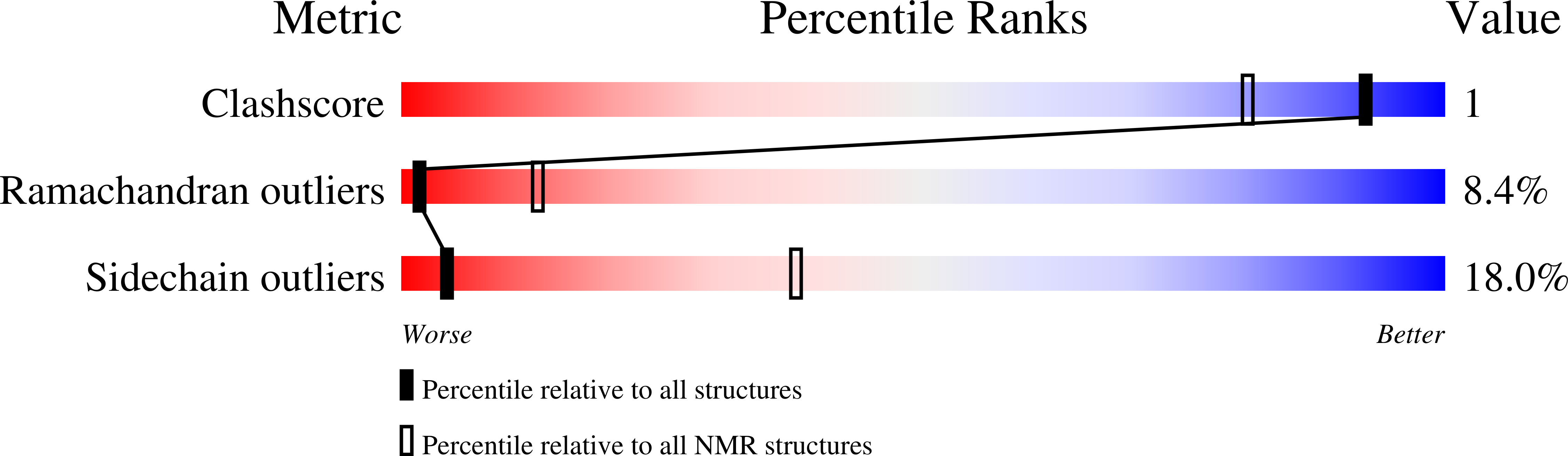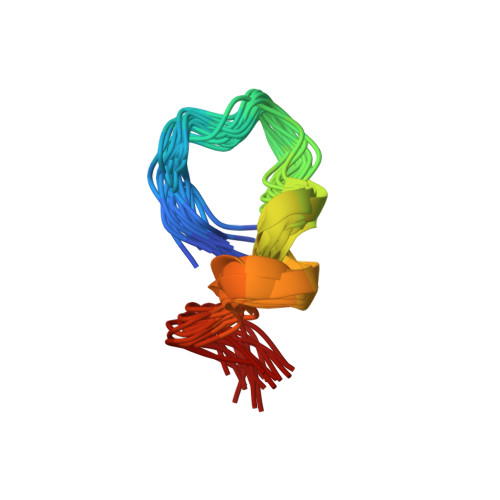Specific recognition between surface loop 2 (132-143) and helix 1 (144-154) within sheep prion protein from in vitro studies of synthetic peptides
Kozin, S.A., Lepage, C., Hui Bon Hoa, G., Rabesona, H., Mazur, A.K., Blond, A., Cheminant, M., Haertle, T., Debey, P., Rebuffat, S.To be published.