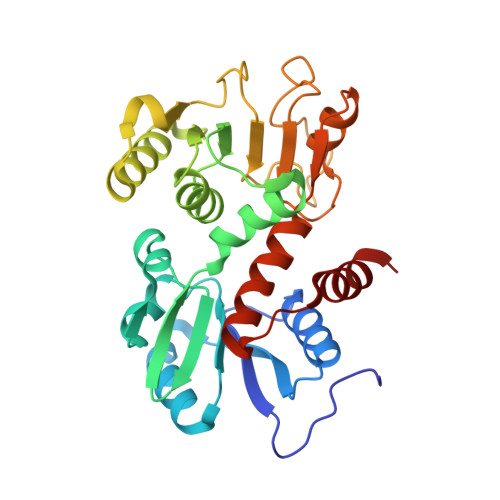
Crystal structure of shikimate 5-dehydrogenase (SDH) bound to NADP: insights into function and evolution
Padyana, A.K., Burley, S.K.(2003) Structure 11: 1005-1013
- PubMed: 12906831 Search on PubMed
- DOI: https://doi.org/10.1016/s0969-2126(03)00159-x
- Primary Citation Related Structures:
1NVT - PubMed Abstract:
The crystal structure of Methanococcus jannaschii shikimate 5-dehydrogenase (MjSDH) bound to the cofactor nicotinamide adenine dinucleotide phosphate (NADP) has been determined at 2.35 A resolution. Shikimate 5-dehydrogenase (SDH) is responsible for NADP-dependent catalysis of the fourth step in shikimate biosynthesis, which is essential for aromatic amino acid metabolism in bacteria, microbial eukaryotes, and plants. The structure of MjSDH is a compact alpha/beta sandwich with two distinct domains, responsible for binding substrate and the NADP cofactor, respectively. A phylogenetically conserved deep cleft on the protein surface corresponds to the enzyme active site. The structure reveals a topologically new domain fold within the N-terminal segment of the polypeptide chain, which binds substrate and supports dimerization. Insights gained from homology modeling and sequence/structure comparisons suggest that the SDHs represent a unique class of dehydrogenases. The structure provides a framework for further investigation to discover and develop novel inhibitors targeting this essential enzyme.
- Laboratories of Molecular Biophysics, The Rockefeller University, 1230 York Avenue, New York, NY 10021, USA.
Organizational Affiliation: