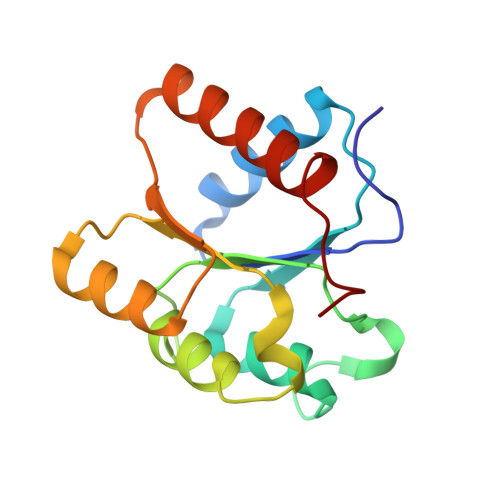
Crystal structures of two cyanobacterial response regulators in apo- and phosphorylated form reveal a novel dimerization motif of phytochrome-associated response regulators
Benda, C., Scheufler, C., Tandeau de Marsac, N., Gaertner, W.(2004) Biophys J 87: 476-487
- PubMed: 15240481 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1529/biophysj.103.033696
- Primary Citation Related Structures:
1K66, 1K68 - PubMed Abstract:
The structures of two response regulators (RRs) from the cyanobacterium Calothrix PCC7601, RcpA and RcpB, were solved to 1.9- and 1.75-A resolution, respectively. RcpA was found in phosphorylated and RcpB in nonphosphorylated form. Both RRs are members of phytochrome-associated, light-sensing two-component signal transduction pathways, based on histidine kinase-mediated receptor autophosphorylation and phosphorelay to a RR. Despite the overall folding similarity to CheY-type RRs ((beta/alpha)(5)-motif), RcpA and RcpB form homodimers, irrespective of their phosphorylation state, giving insight into a signal transduction putatively different from that of other known RRs. Dimerization is accomplished by a C-terminal extension of the RR polypeptide chain, and the surface formed by H4, beta 5, and H5, which constitute a hydrophobic contact area with distinct interactions between residues of either subunit. Sequence alignments reveal that the identified dimerization motif is archetypal for phytochrome-associated RRs, making them a novel subgroup of CheY-type RRs. The protein structures of RcpA and RcpB are compared to the recently presented protein structure of Rcp1 from Synechocystis.
- Max-Planck-Institut für Biochemie, D-82152 Martinsried, Germany.
Organizational Affiliation: