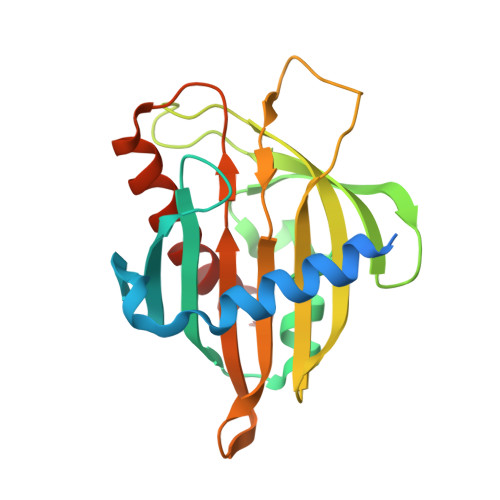
Crystal structure of the Mus musculus cholesterol-regulated START protein 4 (StarD4) containing a StAR-related lipid transfer domain.
Romanowski, M.J., Soccio, R.E., Breslow, J.L., Burley, S.K.(2002) Proc Natl Acad Sci U S A 99: 6949-6954
- PubMed: 12011453 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.052140699
- Primary Citation Related Structures:
1JSS - PubMed Abstract:
The x-ray structure of the mouse cholesterol-regulated START protein 4 (StarD4) has been determined at 2.2-A resolution, revealing a compact alpha/beta structure related to the START domain present in the cytoplasmic C-terminal portion of human MLN64. The volume of the putative lipid-binding tunnel was estimated at 847 A(3), which is consistent with the binding of one cholesterol-size lipid molecule. Comparison of the tunnel-lining residues in StarD4 and MLN64-START permitted identification of possible lipid specificity determinants in both molecular tunnels. Homology modeling of related proteins, and comparison of the StarD4 and MLN64-START structures, showed that StarD4 is a member of a large START domain superfamily characterized by the helix-grip fold. Additional mechanistic and evolutionary studies should be facilitated by the availability of a second START domain structure from a distant relative of MLN64.
- Laboratories of Molecular Biophysics, The Rockefeller University, 1230 York Avenue, New York, NY 10021, USA. romanom@mail.rockefeller.edu
Organizational Affiliation: