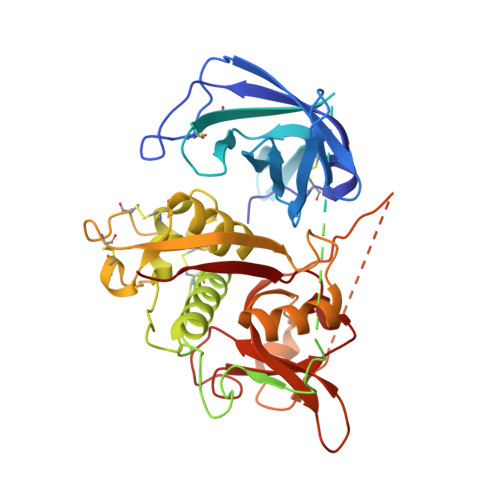
Tetrameric dipeptidyl peptidase I directs substrate specificity by use of the residual pro-part domain
Olsen, J.G., Kadziola, A., Lauritzen, C., Pedersen, J., Larsen, S., Dahl, S.W.(2001) FEBS Lett 506: 201-206
- PubMed: 11602245 Search on PubMed
- DOI: https://doi.org/10.1016/s0014-5793(01)02911-8
- Primary Citation Related Structures:
1JQP - PubMed Abstract:
The crystal structure of mature dipeptidyl peptidase I reveals insight into the unique tetrameric structure, substrate binding and activation of this atypical papain family peptidase. Each subunit is composed of three peptides. The heavy and light chains form the catalytic domain, which adopts the papain fold. The residual pro-part forms a beta-barrel with the carboxylate group of Asp1 pointing towards the substrate amino-terminus. The tetrameric structure appears to stabilize the association of the two domains and encloses a 12700 A3 spherical cavity. The tetramer contains six chloride ions, one buried in each S2 pocket and two at subunit interfaces.
- Center for Crystallographic Studies, University of Copenhagen, Denmark.
Organizational Affiliation: