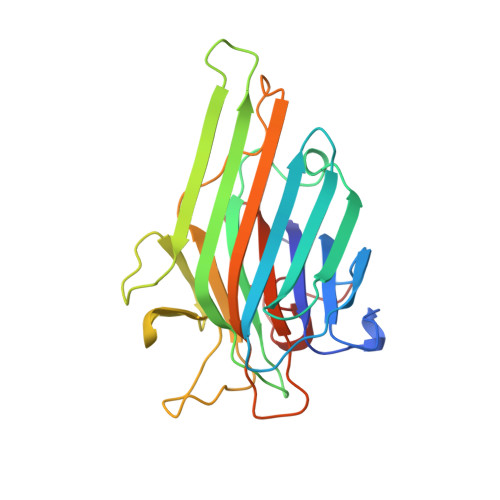
Atomic resolution structure of concanavalin A at 120 K.
Parkin, S., Rupp, B., Hope, H.(1996) Acta Crystallogr D Biol Crystallogr 52: 1161-1168
- PubMed: 15299577 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444996009237
- Primary Citation Related Structures:
1JBC - PubMed Abstract:
The structure of native concanavalin A has been refined to a resolution of 1.2 A against data collected at 120 K. The space group is I222, with a = 61.954 (8), b = 86.053 (11), c = 89.079 (11) A. The structure was refined by restrained weighted least-squares minimization of sum w(F(o)(2) - F(c)(2)(2) with SHELXL92/3/6. The final model contains all of the atoms from 237 amino acids, two metal ions and 271 water molecules spread over 287 sites. Disorder is modelled over two conformations for 30 amino-acid side chains. The final weighted R index on F(2) (wR(2)) on all data was 30.4%. Conventional R indices based on F were 14.2 and 11.8% for all data and for data with F > 4sigma(F), respectively.
- Biology and Biotechnology Research Program, Lawrence Livermore National Laboratory, CA 94550, USA. sp@oedipus.llnl.gov
Organizational Affiliation: