Biophysical characterization of interactions involving importin-alpha during nuclear import.
Catimel, B., Teh, T., Fontes, M.R., Jennings, I.G., Jans, D.A., Howlett, G.J., Nice, E.C., Kobe, B.(2001) J Biological Chem 276: 34189-34198
- PubMed: 11448961 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M103531200
- Primary Citation Related Structures:
1IQ1 - PubMed Abstract:
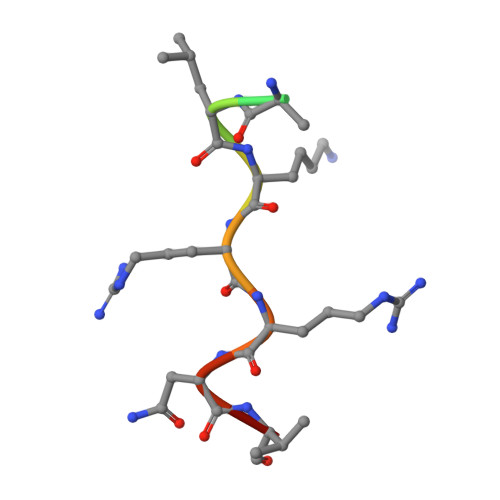
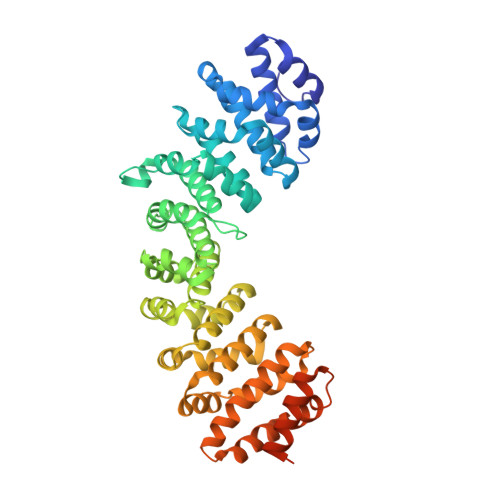
Proteins containing the classical nuclear localization sequences (NLSs) are imported into the nucleus by the importin-alpha/beta heterodimer. Importin-alpha contains the NLS binding site, whereas importin-beta mediates the translocation through the nuclear pore. We characterized the interactions involving importin-alpha during nuclear import using a combination of biophysical techniques (biosensor, crystallography, sedimentation equilibrium, electrophoresis, and circular dichroism). Importin-alpha is shown to exist in a monomeric autoinhibited state (association with NLSs undetectable by biosensor). Association with importin-beta (stoichiometry, 1:1; K(D) = 1.1 x 10(-8) m) increases the affinity for NLSs; the importin-alpha/beta complex binds representative monopartite NLS (simian virus 40 large T-antigen) and bipartite NLS (nucleoplasmin) with affinities (K(D) = 3.5 x 10(-8) m and 4.8 x 10(-8) m, respectively) comparable with those of a truncated importin-alpha lacking the autoinhibitory domain (T-antigen NLS, K(D) = 1.7 x 10(-8) m; nucleoplasmin NLS, K(D) = 1.4 x 10(-8) m). The autoinhibitory domain (as a separate peptide) binds the truncated importin-alpha, and the crystal structure of the complex resembles the structure of full-length importin-alpha. Our results support the model of regulation of nuclear import mediated by the intrasteric autoregulatory sequence of importin-alpha and provide a quantitative description of the binding and regulatory steps during nuclear import.
- Ludwig Institute for Cancer Research, Royal Melbourne Hospital, Victoria 3050, Australia.
Organizational Affiliation: