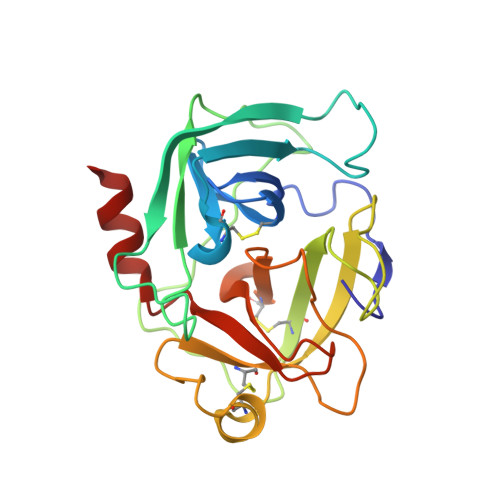
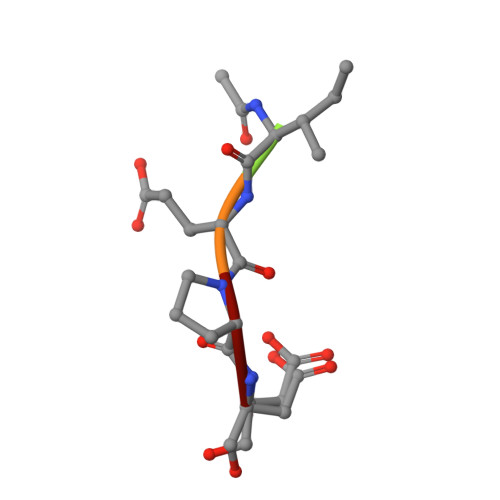
The three-dimensional structure of human granzyme B compared to caspase-3, key mediators of cell death with cleavage specificity for aspartic acid in P1.
Rotonda, J., Garcia-Calvo, M., Bull, H.G., Geissler, W.M., McKeever, B.M., Willoughby, C.A., Thornberry, N.A., Becker, J.W.(2001) Chem Biol 8: 357-368
- PubMed: 11325591 Search on PubMed
- DOI: https://doi.org/10.1016/s1074-5521(01)00018-7
- Primary Citation Related Structures:
1IAU - PubMed Abstract:
Granzyme B, one of the most abundant granzymes in cytotoxic T-lymphocyte (CTL) granules, and members of the caspase (cysteine aspartyl proteinases) family have a unique cleavage specificity for aspartic acid in P1 and play critical roles in the biochemical events that culminate in cell death. We have determined the three-dimensional structure of the complex of the human granzyme B with a potent tetrapeptide aldehyde inhibitor. The Asp-specific S1 subsite of human granzyme B is significantly larger and less charged than the corresponding Asp-specific site in the apoptosis-promoting caspases, and also larger than the corresponding subsite in rat granzyme B. The above differences account for the variation in substrate specificity among granzyme B, other serine proteases and the caspases, and enable the design of specific inhibitors that can probe the physiological functions of these proteins and the disease states with which they are associated.
- Department of Endocrinology and Chemical Biology, Merck Research Laboratories, Rahway, NJ 07065-0900, USA. rotonda@merck.com
Organizational Affiliation: