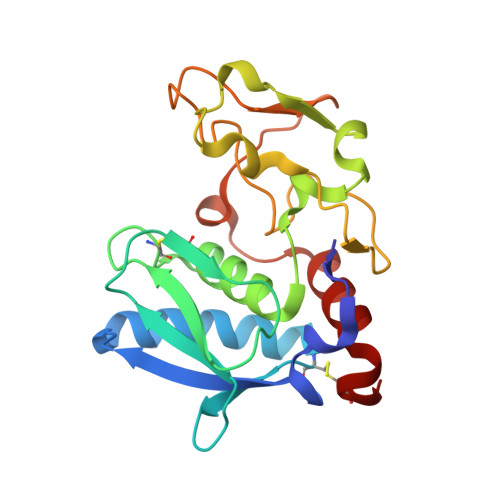
Crystal structures, spectroscopic features, and catalytic properties of cobalt(II), copper(II), nickel(II), and mercury(II) derivatives of the zinc endopeptidase astacin. A correlation of structure and proteolytic activity.
Gomis-Ruth, F.X., Grams, F., Yiallouros, I., Nar, H., Kusthardt, U., Zwilling, R., Bode, W., Stocker, W.(1994) J Biological Chem 269: 17111-17117
- PubMed: 8006015 Search on PubMed
- Primary Citation Related Structures:
1IAA, 1IAB, 1IAE - PubMed Abstract:
The catalytic zinc ion of astacin, a prototypical metalloproteinase from crayfish, has been substituted by Co(II), Cu(II), Hg(II), and Ni(II) in order to probe the role of the metal for both catalysis and structure. Compared to Zn(II)-astacin, Co(II)- and Cu(II)-astacin display enzymatic activities of about 140 and 37%, respectively, while Ni(II)- and Hg(II)-astacin are almost inactive. The electron paramagnetic resonance spectrum of Cu(II)-astacin is typical of 5-fold coordinated copper(II), and its intense absorption maxima at 445 and 325 nm are probably due to ligand-metal charge-transfer transitions involving Tyr-149. This residue had been identified previously by x-ray crystallography of the zinc enzyme as a zinc ligand, in addition to three imidazoles and a glutamic acid-bound water molecule. We present now the refined high-resolution x-ray crystal structures of Cu(II)-, Co(II)-, and Ni(II)-astacin, which exhibit a virtually identical protein framework to the previously analyzed structures of Zn(II)-, apo-, and Hg(II)-astacin. In Co(II)- and Cu(II)-astacin, the metal is penta-coordinated similarly to the native zinc enzyme. In the Ni(II) derivative, however, an additional solvent molecule expands the metal coordination sphere to a distorted octahedral ligand geometry, while in Hg(II)-astacin, no ordered solvent molecule at all is observed in the inner coordination sphere of the metal. This indicates a close correlation between catalytic properties and ground-state metal coordination of astacin.
- Max-Planck-Institut für Biochemie, Martinsried, Germany.
Organizational Affiliation: