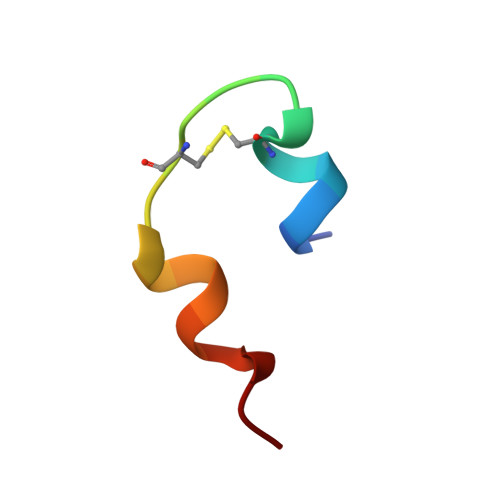
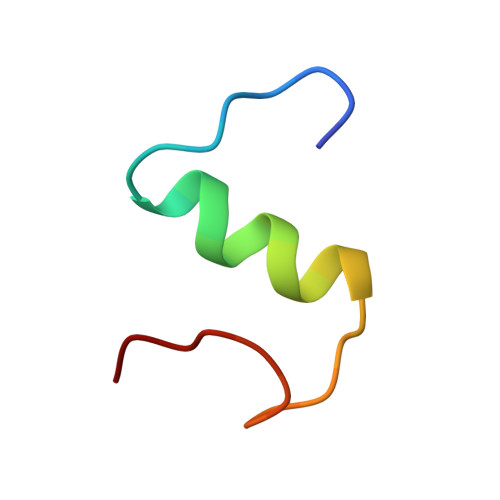
A structural switch in a mutant insulin exposes key residues for receptor binding.
Ludvigsen, S., Olsen, H.B., Kaarsholm, N.C.(1998) J Mol Biology 279: 1-7
- PubMed: 9636695 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1998.1801
- Primary Citation Related Structures:
1A7F - PubMed Abstract:
Despite years of effort to clarify the structural basis of insulin receptor binding no clear consensus has emerged. It is generally believed that insulin receptor binding is accompanied by some degree of conformational change in the carboxy-terminal of the insulin B-chain. In particular, while most substitutions for PheB24 lead to inactive species, glycine or D-amino acids are well tolerated in this position. Here we assess the conformation change by solving the solution structure of the biologically active (GluB16, GlyB24, desB30)-insulin mutant. The structure in aqueous solution at pH 8 reveals a subtle, albeit well-defined rearrangement of the C-terminal decapeptide involving a perturbation of the B20-23 turn, which allows the PheB25 residue to occupy the position normally taken up by PheB24 in native insulin. The new protein surface exposed rationalizes the receptor binding properties of a series of insulin analogs. We suggest that the structural switch is forced by the structure of the underlying core of species invariant residues and that an analogous rearrangement of the C-terminal of the B-chain occurs in native insulin on binding to its receptor.
- Novo Nordisk A/S, Bagsvaerd, Denmark.
Organizational Affiliation: