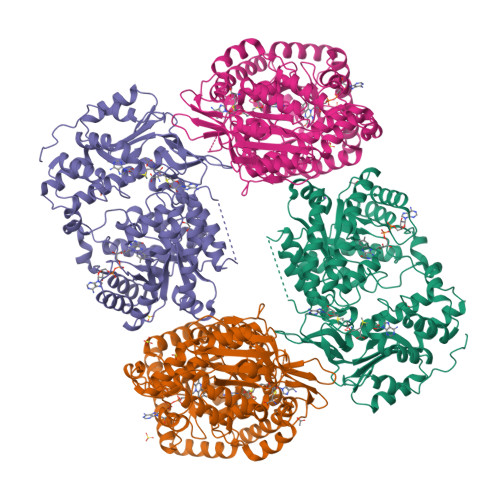
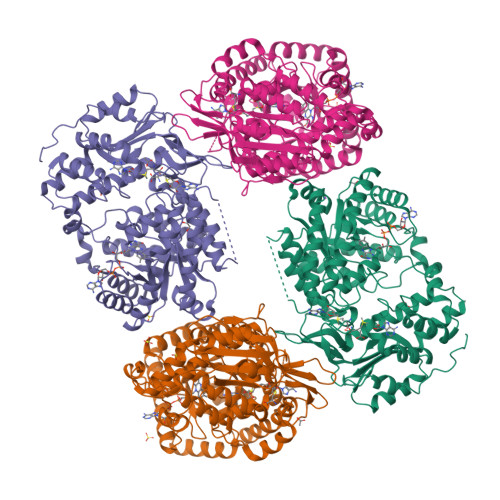
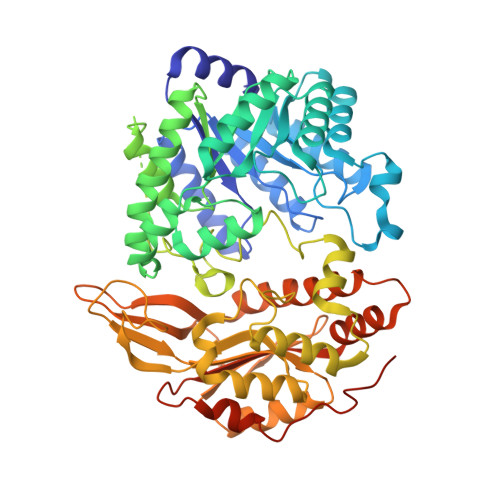
Structural basis of S-adenosylmethionine-dependent allosteric transition from active to inactive states in methylenetetrahydrofolate reductase.
Yamada, K., Mendoza, J., Koutmos, M.(2024) Nat Commun 15: 5167-5167
- PubMed: 38886362
- DOI: https://doi.org/10.1038/s41467-024-49327-5
- Primary Citation of Related Structures:
8UY1, 8UY2 - PubMed Abstract:
Methylenetetrahydrofolate reductase (MTHFR) is a pivotal flavoprotein connecting the folate and methionine methyl cycles, catalyzing the conversion of methylenetetrahydrofolate to methyltetrahydrofolate. Human MTHFR (hMTHFR) undergoes elaborate allosteric regulation involving protein phosphorylation and S-adenosylmethionine (AdoMet)-dependent inhibition, though other factors such as subunit orientation and FAD status remain understudied due to the lack of a functional structural model. Here, we report crystal structures of Chaetomium thermophilum MTHFR (cMTHFR) in both active (R) and inhibited (T) states. We reveal FAD occlusion by Tyr361 in the T-state, which prevents substrate interaction. Remarkably, the inhibited form of cMTHFR accommodates two AdoMet molecules per subunit. In addition, we conducted a detailed investigation of the phosphorylation sites in hMTHFR, three of which were previously unidentified. Based on the structural framework provided by our cMTHFR model, we propose a possible mechanism to explain the allosteric structural transition of MTHFR, including the impact of phosphorylation on AdoMet-dependent inhibition.
Organizational Affiliation:
Department of Chemistry, University of Michigan, Ann Arbor, MI, 48109, USA. yamadak@umich.edu.