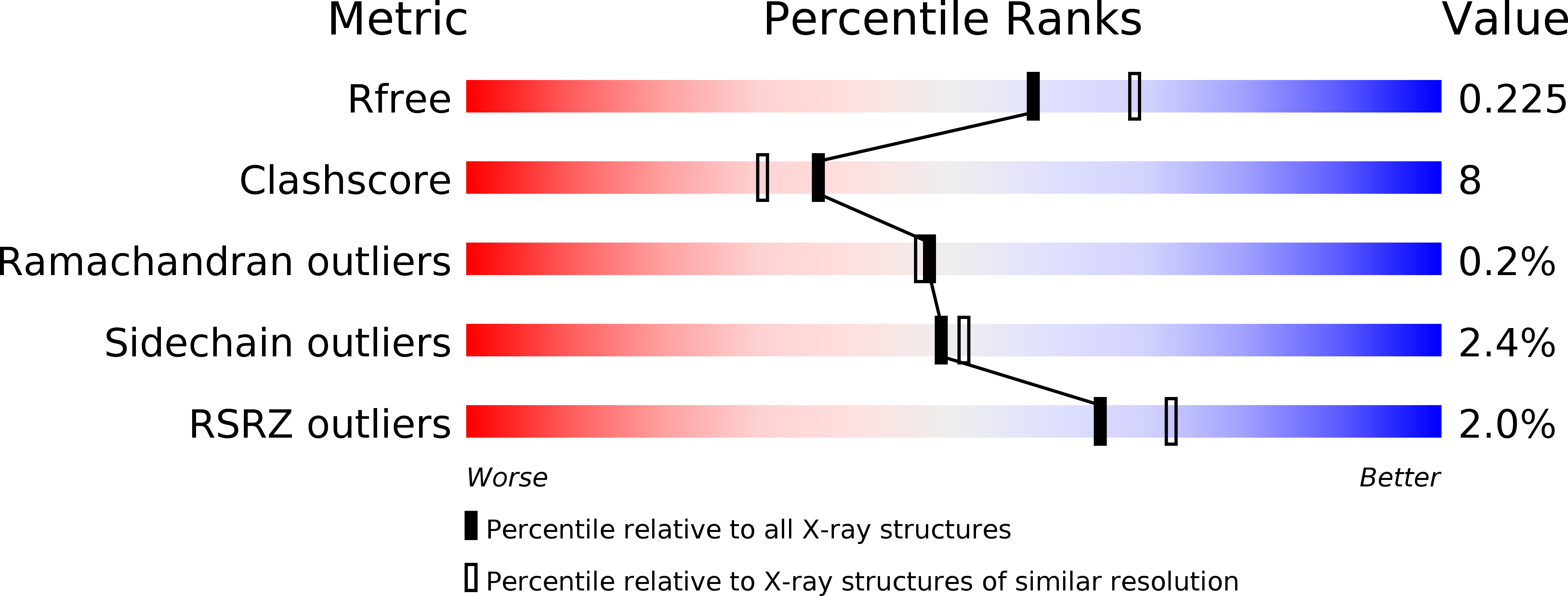
Ligand-binding and metal-exchange crystallographic studies on shrimp alkaline phosphatase.
de Backer, M.M., McSweeney, S., Lindley, P.F., Hough, E.(2004) Acta Crystallogr D Biol Crystallogr 60: 1555-1561
- PubMed: 15333925
- DOI: https://doi.org/10.1107/S0907444904015628
- Primary Citation of Related Structures:
1SHN, 1SHQ - PubMed Abstract:
Alkaline phosphatases (APs) are homodimeric metalloenzymes that catalyze the hydrolysis and transphosphorylation of phosphate monoesters. Each monomer contains a metal-binding triad that for optimal activity is usually occupied by two zinc ions and one magnesium ion. The recently determined crystal structure of cold-active shrimp alkaline phosphatase (SAP) was, however, fully occupied by zinc ions. This paper describes a metal-exchange experiment in which the zinc ion in one binding site (referred to as the M3 site) is replaced by magnesium. Crystal structures revealed a concomitant structural change: the metal exchange causes movement of a ligating histidine into a conformation in which it does not coordinate to the metal ion. The M3 site is relevant to catalysis: its occupation by magnesium is postulated to favour catalysis and it has been suggested to be a regulatory site for other APs. Further crystallographic studies show that ligand binding can induce a conformational change of an active-site arginine from a 'non-docked' (non-interacting) to a 'docked' conformation (interacting with the ligand). The first conformation has only been observed in SAP, while the latter is common in available AP structures. The observation that the arginine does not always bind the substrate may explain the increased catalytic efficiency that is generally observed for cold-active enzymes.
Organizational Affiliation:
European Synchrotron Radiation Facility, France.