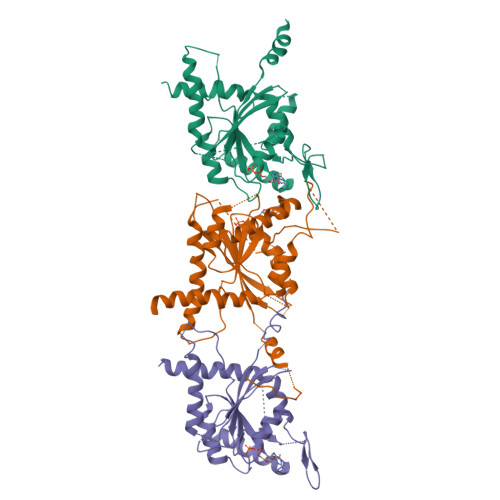
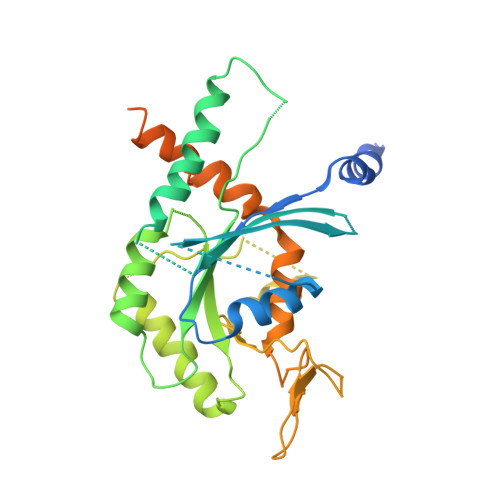
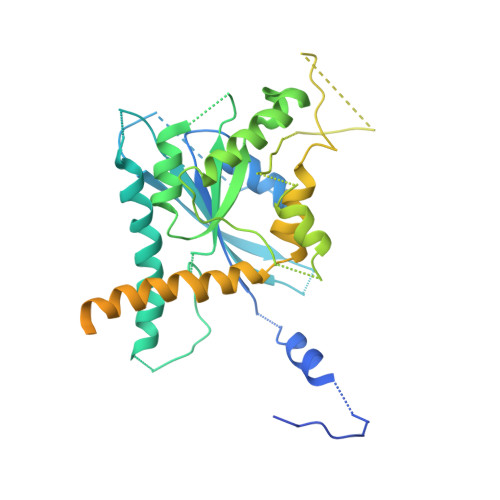
Structural insight into filament formation by mammalian septins.
Sirajuddin, M., Farkasovsky, M., Hauer, F., Kuhlmann, D., Macara, I.G., Weyand, M., Stark, H., Wittinghofer, A.(2007) Nature 449: 311-315
- PubMed: 17637674
- DOI: https://doi.org/10.1038/nature06052
- Primary Citation of Related Structures:
2QA5, 2QAG - PubMed Abstract:
Septins are GTP-binding proteins that assemble into homo- and hetero-oligomers and filaments. Although they have key roles in various cellular processes, little is known concerning the structure of septin subunits or the organization and polarity of septin complexes. Here we present the structures of the human SEPT2 G domain and the heterotrimeric human SEPT2-SEPT6-SEPT7 complex. The structures reveal a universal bipolar polymer building block, composed of an extended G domain, which forms oligomers and filaments by conserved interactions between adjacent nucleotide-binding sites and/or the amino- and carboxy-terminal extensions. Unexpectedly, X-ray crystallography and electron microscopy showed that the predicted coiled coils are not involved in or required for complex and/or filament formation. The asymmetrical heterotrimers associate head-to-head to form a hexameric unit that is nonpolarized along the filament axis but is rotationally asymmetrical. The architecture of septin filaments differs fundamentally from that of other cytoskeletal structures.
Organizational Affiliation:
Abteilung Strukturelle Biologie, Max-Planck-Institut für molekulare Physiologie, Otto-Hahn-Strasse 11, 44227 Dortmund, Germany.