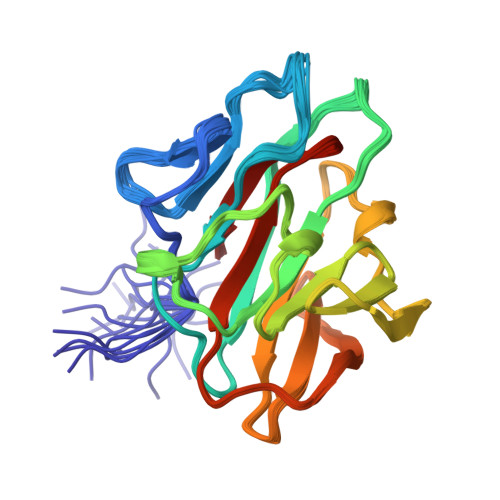
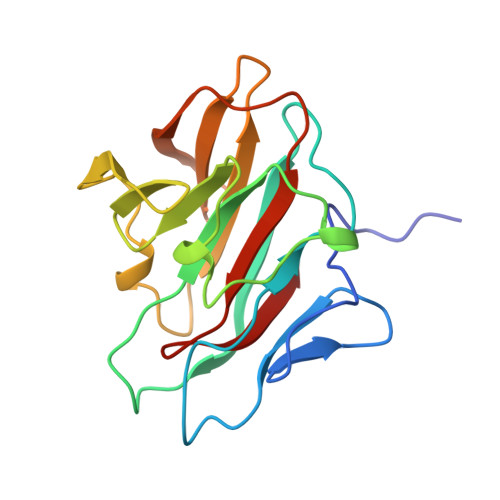
Structural and functional characterization of the NHR1 domain of the Drosophila neuralized E3 ligase in the notch signaling pathway.
He, F., Saito, K., Kobayashi, N., Harada, T., Watanabe, S., Kigawa, T., Guntert, P., Ohara, O., Tanaka, A., Unzai, S., Muto, Y., Yokoyama, S.(2009) J Mol Biology 393: 478-495
- PubMed: 19683535
- DOI: https://doi.org/10.1016/j.jmb.2009.08.020
- Primary Citation of Related Structures:
2E63, 2YUE - PubMed Abstract:
The Notch signaling pathway is critical for many developmental processes and requires complex trafficking of both Notch receptor and its ligands, Delta and Serrate. In Drosophila melanogaster, the endocytosis of Delta in the signal-sending cell is essential for Notch receptor activation. The Neuralized protein from D. melanogaster (Neur) is a ubiquitin E3 ligase, which binds to Delta through its first neuralized homology repeat 1 (NHR1) domain and mediates the ubiquitination of Delta for endocytosis. Tom, a Bearded protein family member, inhibits the Neur-mediated endocytosis through interactions with the NHR1 domain. We have identified the domain boundaries of the novel NHR1 domain, using a screening system based on our cell-free protein synthesis method, and demonstrated that the identified Neur NHR1 domain had binding activity to the 20-residue peptide corresponding to motif 2 of Tom by isothermal titration calorimetry experiments. We also determined the solution structure of the Neur NHR1 domain by heteronuclear NMR methods, using a (15)N/(13)C-labeled sample. The Neur NHR1 domain adopts a characteristic beta-sandwich fold, consisting of a concave five-stranded antiparallel beta-sheet and a convex seven-stranded antiparallel beta-sheet. The long loop (L6) between the beta6 and beta7 strands covers the hydrophobic patch on the concave beta-sheet surface, and the Neur NHR1 domain forms a compact globular fold. Intriguingly, in spite of the slight, but distinct, differences in the topology of the secondary structure elements, the structure of the Neur NHR1 domain is quite similar to those of the B30.2/SPRY domains, which are known to mediate specific protein-protein interactions. Further NMR titration experiments of the Neur NHR1 domain with the 20-residue Tom peptide revealed that the resonances originating from the bottom area of the beta-sandwich (the L3, L5, and L11 loops, as well as the tip of the L6 loop) were affected. In addition, a structural comparison of the Neur NHR1 domain with the first NHR domain of the human KIAA1787 protein, which is from another NHR subfamily and does not bind to the 20-residue Tom peptide, suggested the critical amino acid residues for the interactions between the Neur NHR1 domain and the Tom peptide. The present structural study will shed light on the role of the Neur NHR1 domain in the Notch signaling pathway.
Organizational Affiliation:
RIKEN Systems and Structural Biology Center, Tsurumi-ku, Yokohama, Japan.