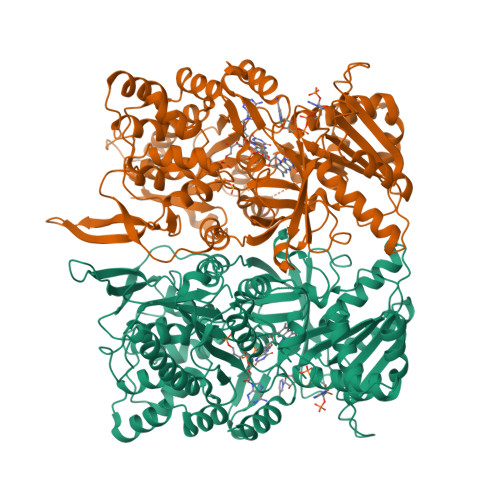
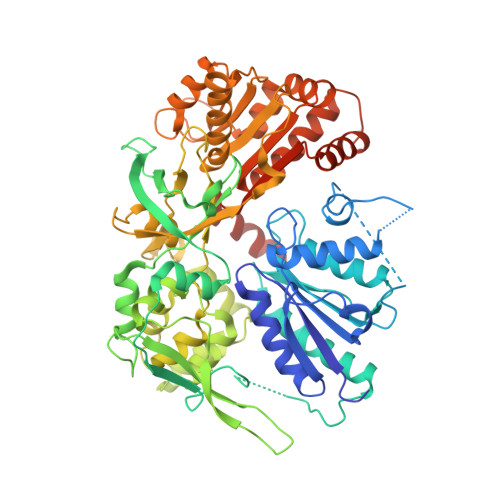
Structural basis for isozyme-specific regulation of electron transfer in nitric-oxide synthase
Garcin, E.D., Bruns, C.M., Lloyd, S.J., Hosfield, D.J., Tiso, M., Gachhui, R., Stuehr, D.J., Tainer, J.A., Getzoff, E.D.(2004) J Biological Chem 279: 37918-37927
- PubMed: 15208315
- DOI: https://doi.org/10.1074/jbc.M406204200
- Primary Citation of Related Structures:
1TLL - PubMed Abstract:
Three nitric-oxide synthase (NOS) isozymes play crucial, but distinct, roles in neurotransmission, vascular homeostasis, and host defense, by catalyzing Ca(2+)/calmodulin-triggered NO synthesis. Here, we address current questions regarding NOS activity and regulation by combining mutagenesis and biochemistry with crystal structure determination of a fully assembled, electron-supplying, neuronal NOS reductase dimer. By integrating these results, we structurally elucidate the unique mechanisms for isozyme-specific regulation of electron transfer in NOS. Our discovery of the autoinhibitory helix, its placement between domains, and striking similarities with canonical calmodulin-binding motifs, support new mechanisms for NOS inhibition. NADPH, isozyme-specific residue Arg(1400), and the C-terminal tail synergistically repress NOS activity by locking the FMN binding domain in an electron-accepting position. Our analyses suggest that calmodulin binding or C-terminal tail phosphorylation frees a large scale swinging motion of the entire FMN domain to deliver electrons to the catalytic module in the holoenzyme.
Organizational Affiliation:
Department of Molecular Biology and the Skaggs Institute for Chemical Biology, The Scripps Research Institute, La Jolla, California 92037, USA. edg@scripps.edu