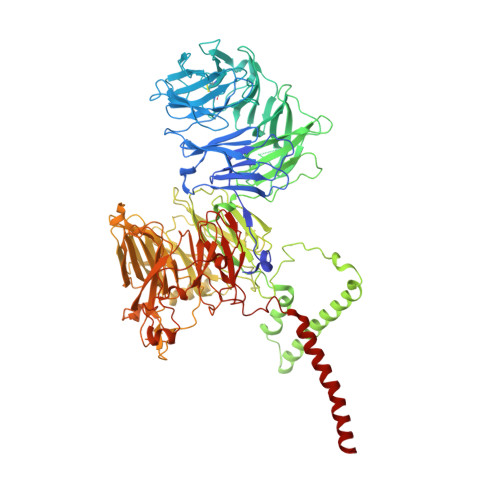
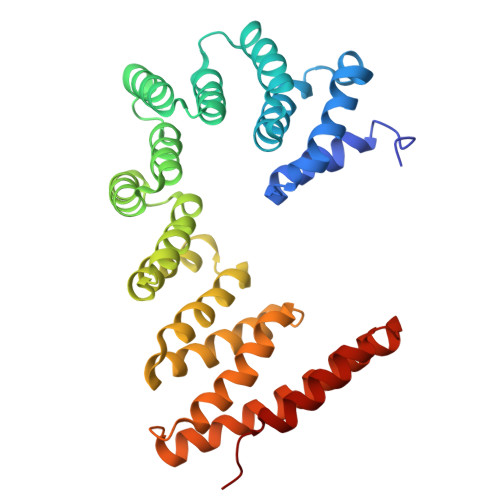
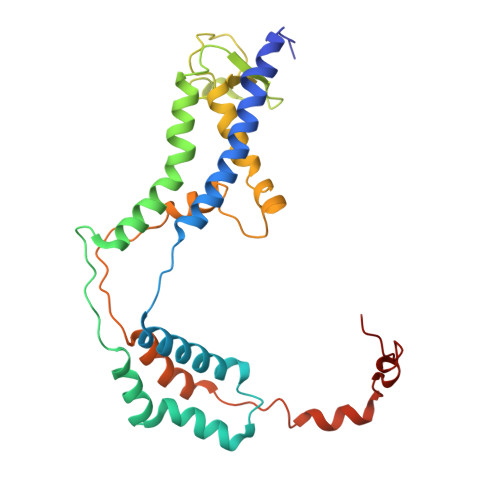
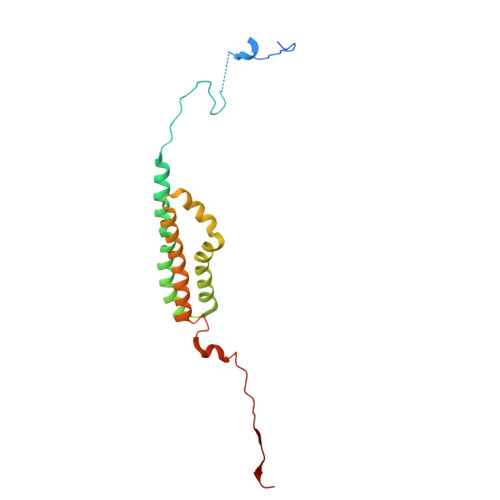
The ER membrane protein complex acts as a chaperone to promote the biogenesis of multi-bundle membrane proteins.
Stanton, M., Singal, B., Biswal, M., Agarwal, M., Scheuing, C.E., Vargas, G.D., Gao, A., Gifford, C.A., Pleiner, T.(2026) bioRxiv
- PubMed: 41648177 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.64898/2026.01.14.699575
- Primary Citation Related Structures:
9ZZ6 - PubMed Abstract:
Nearly half of the ~5,000 human membrane proteins need to assemble into stoichiometric complexes as part of their biogenesis at the endoplasmic reticulum (ER) membrane. How ER resident biogenesis factors coordinate membrane insertion, folding and assembly is not well understood. Here, we demonstrate that the ER membrane protein complex (EMC) insertase additionally acts as a chaperone to facilitate the assembly of heterotrimeric voltage-gated calcium channels (Ca v ). Using function-separating mutations and inhibitory nanobodies we show that nascent Ca v channels are degraded prematurely when EMC's chaperone function is selectively perturbed. Blocking EMC's chaperone function strongly impaired Ca v -dependent cardiomyocyte contraction. EMC engagement of the pore-forming Ca v α-subunit occurred co-translationally and required Ca v α's first transmembrane domain bundle to protrude from the nascent ribosome•Sec61•multipass translocon complex. Our findings establish a chaperone function for the EMC and reveal that biogenesis of multi-bundle membrane proteins requires a highly orchestrated, co-translational interplay between ER biogenesis factors.
- Department of Molecular & Cellular Physiology, Stanford University, Stanford, CA 94305, USA.
Organizational Affiliation: