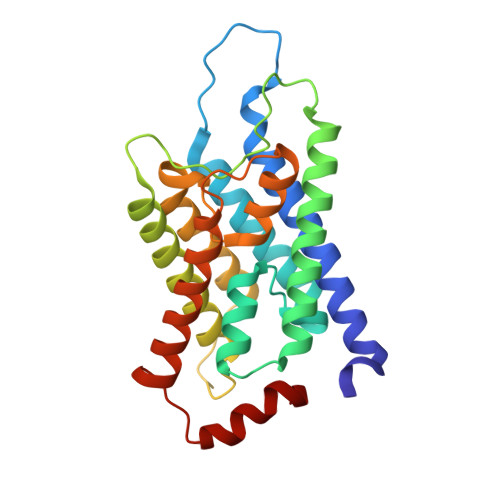
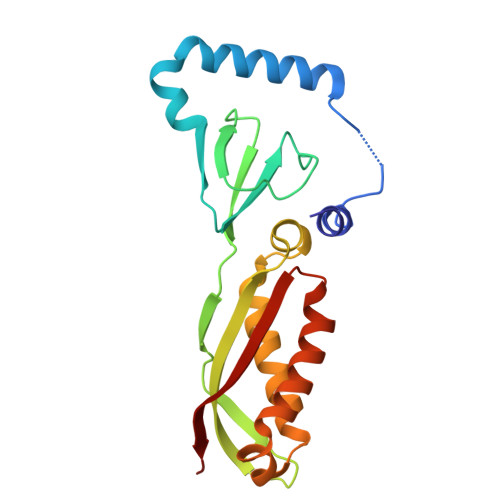
Stomatin encapsulates aquaporin-1 and urea transporter-B in the erythrocyte membrane.
Vallese, F., Li, H., Barazzuol, L., Cali, T., Clarke, O.B.(2026) Sci Adv 12: eaec1721-eaec1721
- PubMed: 41921000 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.aec1721
- Primary Citation Related Structures:
9Z7U, 9ZCZ, 9ZD0, 9ZD2, 9ZD5 - PubMed Abstract:
Stomatin is a ubiquitous and highly expressed protein in erythrocytes, which associates with cholesterol-rich microdomains in the plasma membrane and is known to regulate the activity of multiple ion channels and transporters, but the structural basis of association with stomatin targets remains unknown. Here, we describe high-resolution structures of multiple stomatin complexes with endogenous binding partners isolated from human erythrocyte membranes, revealing that stomatin specifically associates with two membrane proteins involved in water transport and cell volume regulation, aquaporin-1 and the urea transporter SLC14A1. Together, our results reveal the structural basis of stomatin oligomerization, membrane association, and target recruitment and identify a putative role for stomatin in the regulation of osmotic balance in the erythrocyte.
- Structural Biology Initiative, CUNY Advanced Science Research Center, New York, NY 10031, USA.
Organizational Affiliation: