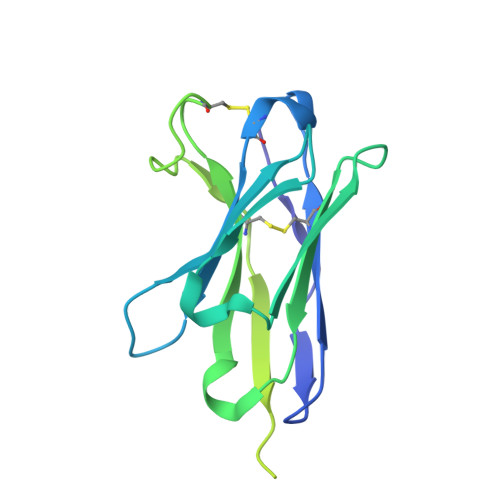
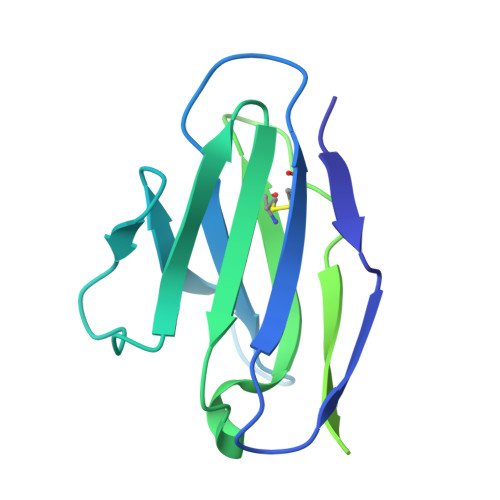
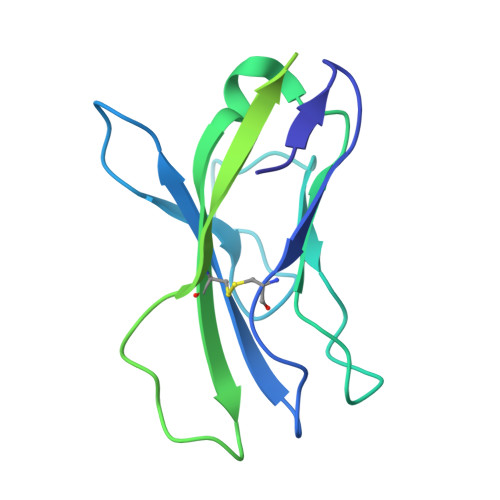
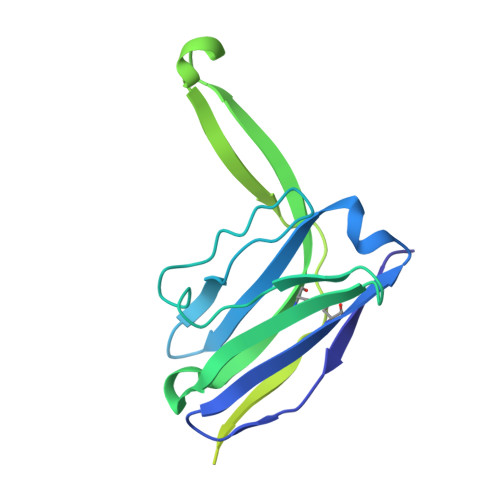
Conformational landscape of HIV-1 Env from closed to fully open.
Cui, J., Lin, Z.J., Ghosh, S., Du, J., Sadeesh, R., Weiner, D.B., Pallesen, J.(2026) Nat Commun
- PubMed: 41735302 Search on PubMed
- DOI: https://doi.org/10.1038/s41467-026-69921-z
- Primary Citation Related Structures:
9NBT, 9NBY, 9NC0, 9NC3, 9NC6, 9NC8, 9OAJ, 9YQO - PubMed Abstract:
The molecular mechanism of HIV-1 entry into host cells is governed by dynamic conformational changes to its envelope glycoprotein (Env), which are triggered by the engagement of the host receptor CD4 and coreceptors. Structural insights into these transitions have been advanced by cryo-electron tomography (cryo-ET), resolving Env structures in closed and multifarious open states within native membranes, and by cryo-electron microscopy (cryo-EM), which has provided atomic details of these states. In this study, we determine cryo-EM structures of soluble native-like Env in complex with antibody 3BC315, antibody b12, CD4, or a combination of 3BC315 and b12, capturing previously uncharacterized conformational states. Observing enhanced 3BC315 binding occupancy in the presence of b12, we investigate the cooperativity of these antibodies using mass photometry and neutralization assays. Integrating these states with the literature, we establish a classification framework for symmetric and asymmetric Env states, categorizing by their degree of openness and stepwise structural rearrangements. Our findings refine the mechanistic understanding of HIV-1 Env dynamics and provide a structural roadmap for targeting dynamic Env states to develop more potent vaccines and immunotherapies.
- Vaccine and Immunotherapy Center, The Wistar Institute, Philadelphia, PA, USA.
Organizational Affiliation: