Solid-phase synthesis and biological evaluation of des-hydroxy pseudouridimycin analogs.
Anwar, A.F., You, L., Degen, D., Burns, K.J., Garza, M.J., Ebright, R.H., Del Valle, J.R.(2026) ACS Med Chem Lett 17: 835-840
- PubMed: 41890570 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acsmedchemlett.5c00684
- Primary Citation Related Structures:
9YNP, 9YNQ - PubMed Abstract:
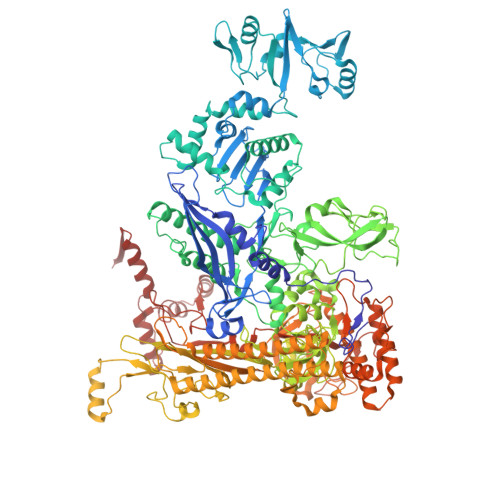
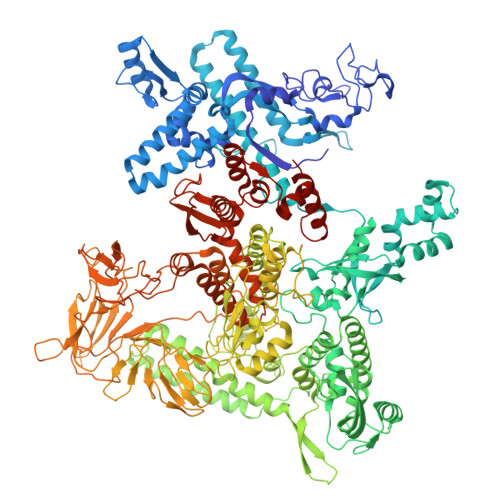
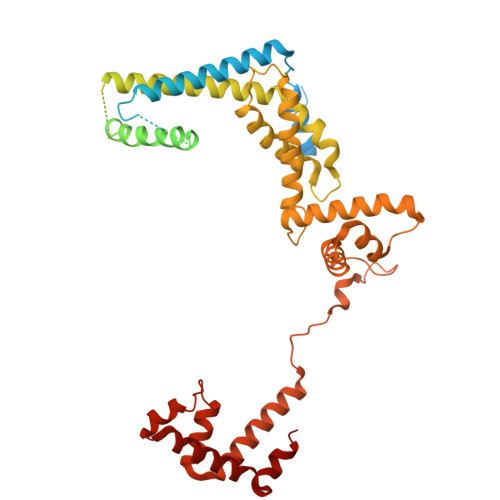
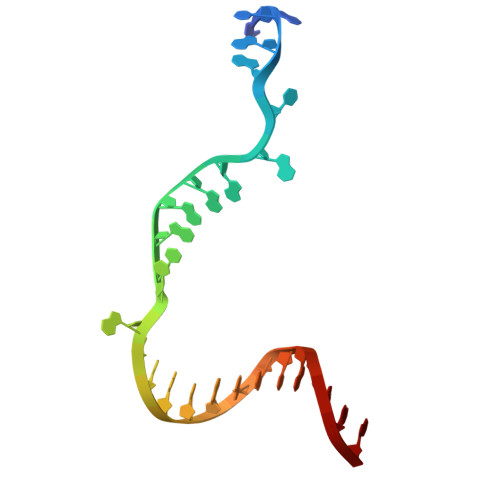
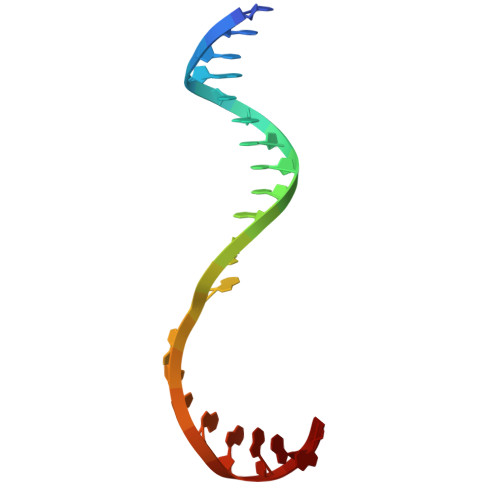
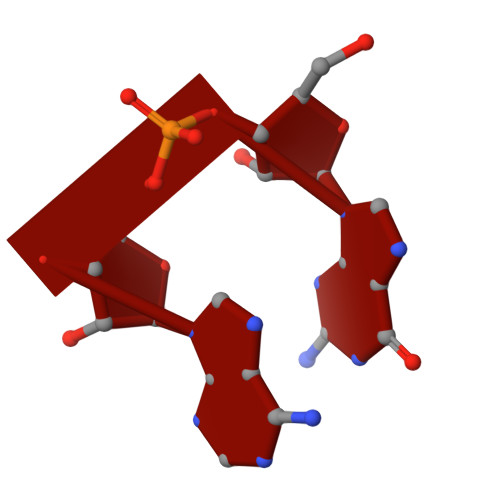
Pseudouridimycin (PUM) is a C-nucleoside/peptide antibiotic that selectively inhibits bacterial RNA polymerase (RNAP) and exhibits potent activity against drug-resistant pathogens. However, PUM suffers from chemical instability due to self-immolative cleavage of its central hydroxamate bond. Here, we employed cryo-electron microscopy to determine structures of PUM ( 1 ) and a chemically stabilized des-hydroxy analog of PUM ( 2a ) bound to an Escherichia coli RNAP transcription complex. Guided by the observed bound conformation, we developed an efficient solid-phase synthesis of 50 des-hydroxy PUM analogs modified at the Gln residue and Gdn-Gly tail. Several analogs retained low-micromolar RNAP-inhibitory activity, with a para -substituted phenyl amidine analog ( 54 ) emerging as the most potent inhibitor (IC 50 = 0.95 μM). These results establish a versatile synthetic platform and structural framework for optimizing stabilized PUM derivatives and provide a foundation for the development of RNAP-targeted therapeutics against resistant bacterial pathogens.
- Department of Chemistry & Biochemistry, University of Notre Dame, Notre Dame, Indiana 46556, United States.
Organizational Affiliation: