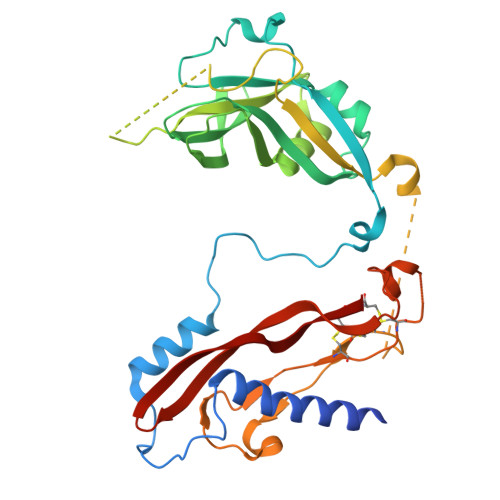
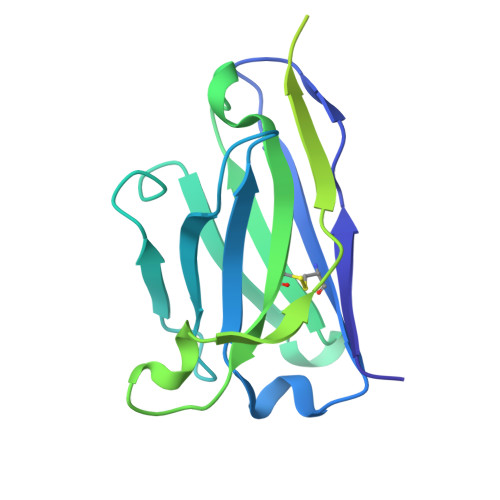
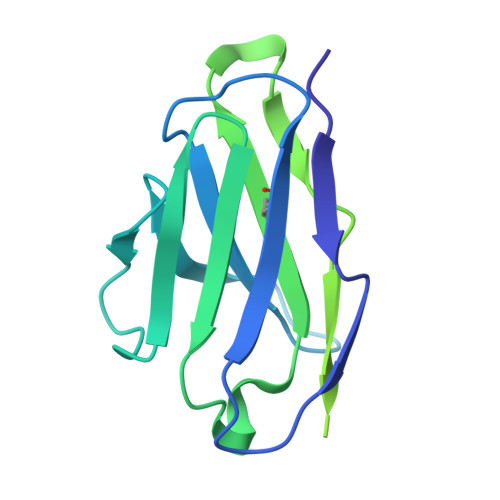
Selective blockade of latent TGF-beta 1 activation suppresses tissue fibrosis with good safety.
Kanamori, M., Sato, I., Koo, C.X., Sun, Y., Kawauchi, H., Nakagawa, K., Murai, A., Asanuma, K., Gan, S.W., Pang, C.L., Shimizu, Y., Shida-Kawazoe, M., Kanamaru, C., Kayukawa, Y., Hada, N., Ohmine, K., Kitazawa, T., Nezu, J., Igawa, T., Shimada, H.(2026) Commun Med (Lond) 6
- PubMed: 41606188 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s43856-026-01408-w
- Primary Citation Related Structures:
9VJJ - PubMed Abstract:
Fibrosis is a hallmark of organ failure observed after chronic epithelial injury and inflammation. The transforming growth factor beta (TGF-β) is the master regulator of fibrogenesis, so blockade of the TGF-β pathway is a potential treatment strategy for fibrosis; however, the therapeutic potential of pan-TGF-β blockade is limited by side effects. We generated SOF10, a humanized antibody that targets latent TGF-β1 and selectively blocks protease- and integrin αvβ8-mediated latent TGF-β1 activation. We conducted gene expression and histological analyses in nonalcoholic steatohepatitis (NASH)/liver fibrosis and renal interstitial fibrosis models. We also evaluated the combination effect of SOF10 with an immune checkpoint inhibitor in a syngeneic mouse model and performed safety studies in mice and monkeys. Here we show that SOF10 reduces fibrosis in NASH/liver fibrosis and renal interstitial fibrosis models and improves renal function in a chronic kidney disease model. Furthermore, the combination of SOF10 with an anti-PD-L1 antibody decreases tumor growth in a syngeneic mouse model. SOF10 demonstrates safety in both mice and monkeys. Selective blockade of latent TGF-β1 activation represents a promising approach for treating a broad range of fibrotic diseases and cancers. By specifically targeting TGF-β1, SOF10 may offer a safer and more effective therapeutic option compared to non-selective TGF-β inhibitors. This strategy has the potential to transform the treatment paradigm for fibrosis-related conditions.
- Research Division, Chugai Pharmaceutical Co. Ltd., Yokohama, Kanagawa, Japan.
Organizational Affiliation: