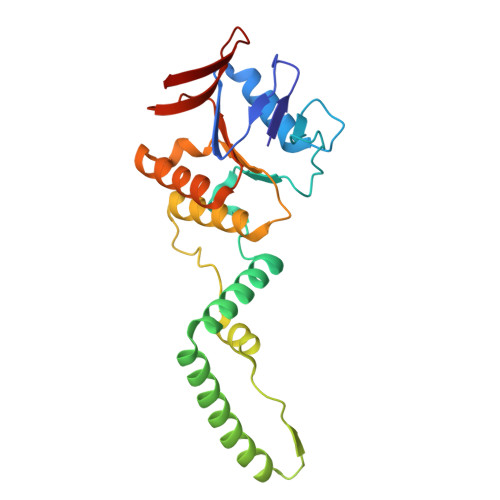
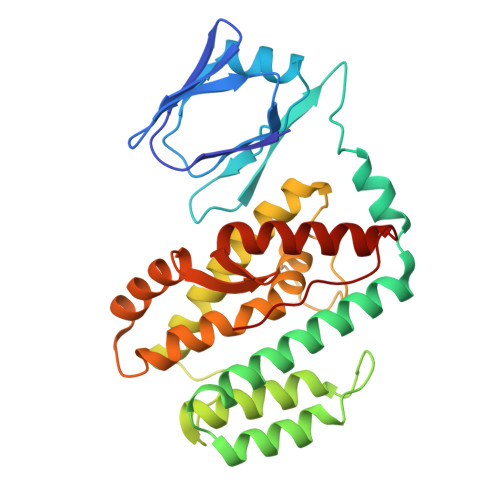
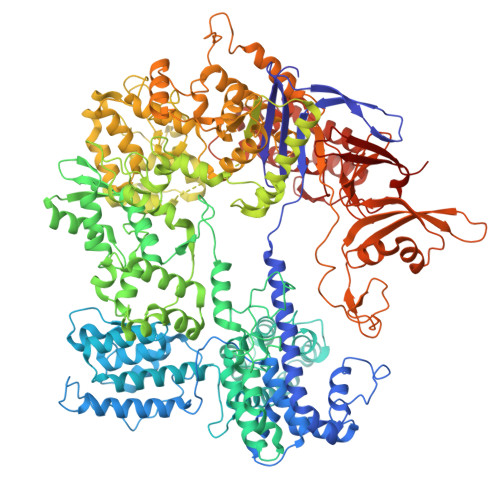
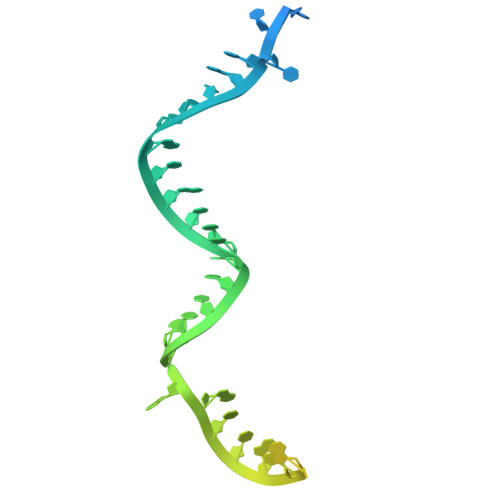
Structural basis for Cas9-directed spacer acquisition in type II-A CRISPR-Cas systems.
Li, Z., Li, Y., Kong, J., Wu, Q., Huang, P., Zhang, Y., Wu, W., Chen, M., Liu, Y., Lin, H., Hou, L., Liu, G., Zeng, T., He, Y., Hu, C., Yang, Z., Lu, M., Luo, M., Xiao, Y.(2026) Mol Cell 86: 805-816.e4
- PubMed: 41702404 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2026.01.024
- Primary Citation Related Structures:
9VJ8, 9VJ9, 9VJA, 9VJB - PubMed Abstract:
CRISPR-Cas systems confer prokaryotic immunity by integrating foreign DNA (prespacers) into host arrays. Type II-A systems employ Cas9 for protospacer-adjacent motif (PAM) recognition and coordinate with Csn2 and the Cas1-Cas2 integrase during spacer acquisition, yet their structural basis remains unresolved. Here, we report cryo-electron microscopy (cryo-EM) structures of the Enterococcus faecalis Cas9-Csn2-Cas1-Cas2 supercomplex in apo and DNA-bound states. The apo state (Cas9₂-Csn2₈-Cas1₈-Cas2₄) is a resting complex, while DNA binding forms a prespacer-catching complex threading DNA through Csn2's channel, enabling Cas9 to interrogate the PAM sequence while sliding along the DNA. Cas9 and Csn2 jointly define a 30-bp DNA segment matching the prespacer length. Cas9 dissociation triggers structural reconfiguration of the Csn2-Cas1-Cas2 assembly. This exposes the PAM-proximal DNA, allowing Cas1-Cas2 to bind the exposed site for subsequent prespacer processing and directional integration. These findings reveal how Cas9, Csn2, and Cas1-Cas2 couple PAM recognition with prespacer selection, ensuring fidelity during adaptation.
- State Key Laboratory of Natural Medicines, School of Pharmacy, China Pharmaceutical University, Nanjing, China; Chongqing Innovation Institute, China Pharmaceutical University, Chongqing 401135, China; Department of Biological Sciences, Faculty of Science, National University of Singapore, Singapore 117543, Singapore.
Organizational Affiliation: