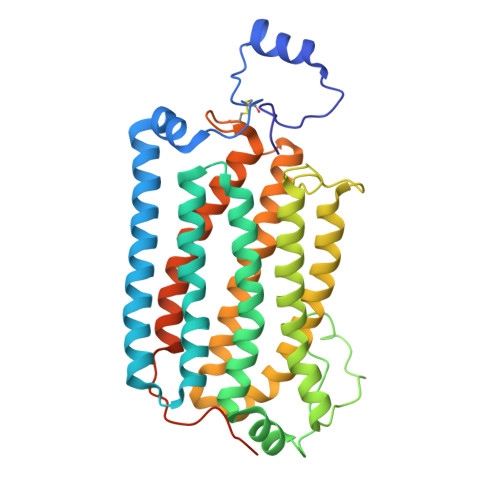
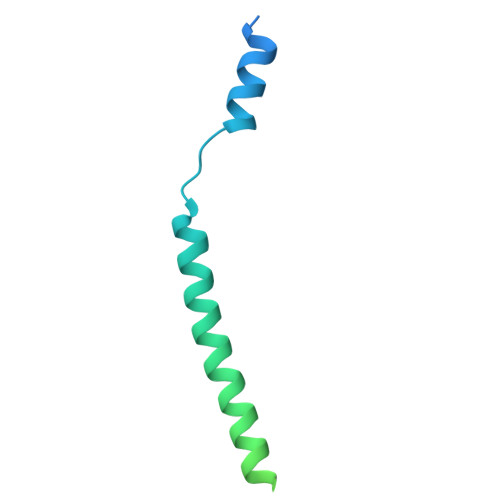
Structures of Ost alpha / beta reveal a unique fold and bile acid transport mechanism.
Yang, X., Cui, N., Li, T., He, X., Zhang, H., Wu, C., Li, Y., Ma, X., Xu, H.E.(2026) Nature 651: 260-267
- PubMed: 41606328 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41586-025-10029-7
- Primary Citation Related Structures:
9UNV, 9UO1, 9UO2 - PubMed Abstract:
Bile acid and steroid hormone homeostasis are critical for human health, with disruptions linked to metabolic and endocrine disorders 1,2 . The organic solute transporter Ostα/β, essential for bile acid efflux in enterohepatic circulation 3 , has long defied mechanistic elucidation. Here we present cryogenic electron microscopy structures of human Ostα/β in apo and substrate-bound states at 2.6-3.1 Å resolution, revealing a distinctive membrane protein architecture that defines a new transporter class. Ostα/β forms a symmetric tetramer of heterodimers, with each Ostα subunit showing a new seven-transmembrane fold, augmented by a single transmembrane helix of Ostβ. This architecture is stabilized by extensive lipid modifications, including a palmitoylated cysteine-rich motif that forms a lateral substrate-binding groove. The structures uncover a unique transport pathway featuring two substrate-binding sites connected by an amphipathic helix-gated conduit. This design, conserved in the evolutionarily related TMEM184 family, suggests an ancient mechanism for substrate translocation. Electrophysiological studies demonstrate voltage-sensitive, bidirectional transport driven by electrochemical gradients, elucidating the efflux role of Ostα/β in vivo. Lipid interactions, notably palmitoylation-dependent trafficking, emerge as critical for stability and function. These findings clarify the molecular mechanism of Ostα/β, provide a structural basis for disease-associated mutations 4,5 and establish a paradigm for lipid-modified membrane transport.
- Research Center for Medicinal Structural Biology, National Research Center for Translational Medicine at Shanghai, State Key Laboratory of Medical Genomics, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China.
Organizational Affiliation: