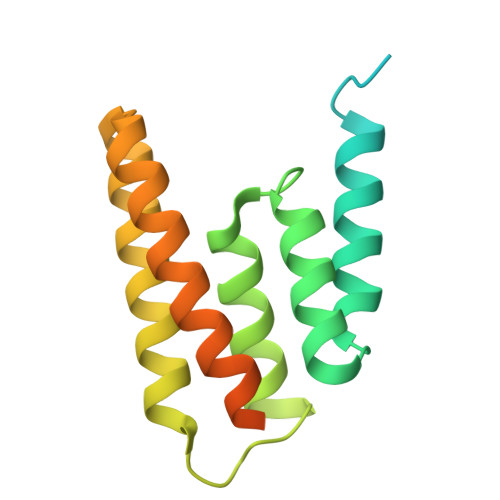
Architecture of surface tubular element of poxvirus.
Yu, F., Jin, G., Liu, Y., Liu, Z., Yao, J., Wang, J., Xie, D., Rao, Z., Yan, L., Zhang, Y., Sun, Z., Lou, Z.(2026) mBio 17: e0314325-e0314325
- PubMed: 41778992 Search on PubMed
- DOI: https://doi.org/10.1128/mbio.03143-25
- Primary Citation Related Structures:
9U9H - PubMed Abstract:
Poxviruses are large enveloped DNA viruses that cause severe human infectious diseases. The mature virion of poxvirus is covered with dense surface tubular elements (STEs), which play a role in assembly progress of mature virions (MVs) and inhibit host cell protein synthesis. However, the composition and assembly of STEs remain unclear. Cryo-electron microscopy (cryo-EM) has proven to be a powerful technique for determining the structure of proteins from complex biological samples. By integrating high-resolution cryo-EM maps with mass spectrometry, we reveal that STEs are helically assembled from two transmembrane proteins, A14 and A17, which bind to phospholipid molecules and form the tubular scaffold along the poxviral membrane. Extensive intermolecular interactions, including A14 dimers and A14-A17 complexes, drive the remarkable structural stability of STEs. Structural analysis further emphasizes the reticulon-like properties of A17, which promote membrane curvature and stabilize the tubular architecture. These results provide novel insights into the STE assembly, morphogenesis, and surface organization of poxviruses, offering valuable information for the development of vaccines and antiviral strategies against poxvirus infections.IMPORTANCESurface tubular elements (STEs) are critical components of poxvirus mature virions and play a role in suppressing host cell protein synthesis. In this study, we isolated and purified STEs from native poxvirus virions and subsequently determined their core composition and high-resolution architecture. We identified that STE is mainly composed of membrane proteins A14 and A17, along with phospholipid molecules. Within the repeat structural unit of STE, A14 proteins form two homodimers within the repeating unit, with A17 monomers flanking either side. Phospholipid molecules are distributed within the A14-A14 and A14-A17 interfaces. Our study not only revealed the molecular structures of A14 and A17 but also further emphasized that the reticulon-like and highly oligomerized characteristics of A17 provide membrane curvature, while the A14-A17-phospholipid network stabilizes the tubular structure. We proposed a hypothetical model that A17 drives changes in viral membrane curvature during maturation. These findings enhance our understanding of poxvirus biology and may guide therapeutic strategies against poxvirus infections.
- MOE Key Laboratory of Protein Science, School of Basic Medical Sciences, Tsinghua University, Beijing, China.
Organizational Affiliation: