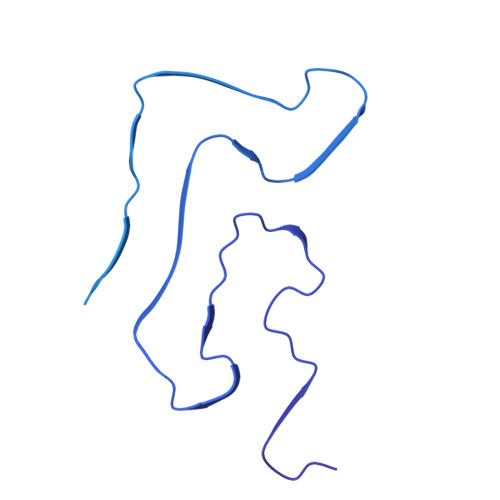
Distinct TAF15 amyloid filament folds define multiple subtypes of FTLD-TAF15.
Tetter, S., Varghese, N.R., Murzin, A.G., De Coster, W., Van den Broeck, M., Roeber, S., Joseph, J.T., Newell, K., Castellani, R., Das, S., Ang, L.C., Synofzik, M., Herms, J., Rademakers, R., Ghetti, B., Lashley, T., Mackenzie, I.R.A., Neumann, M., Ryskeldi-Falcon, B.(2026) bioRxiv
- PubMed: 41648099 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.64898/2026.01.12.698957
- Primary Citation Related Structures:
9THM, 9THN, 9THP, 9TKE, 9TKF, 9TKG, 9TKH, 9TKI, 9TKJ, 9TKK, 9TKL, 9TKZ, 9TL2 - PubMed Abstract:
Neurodegenerative diseases are characterised by the assembly of a limited number of disease-specific proteins into amyloid filaments, which form intracellular inclusions or extracellular deposits in the central nervous system (CNS) 1,2 . We previously found that amyloid filaments of TATA-binding protein-associated factor 15 (TAF15) characterise a subtype of frontotemporal lobar degeneration with FET protein-immunoreactive inclusions (FTLD-FET) 3 , termed atypical FTLD with ubiquitin-positive inclusions (aFTLD-U) 4 , which causes early-onset, rapidly progressive behavioural variant frontotemporal dementia (FTD). However, it was not clear if TAF15 proteinopathy was more widespread in neurodegenerative diseases. Two additional FTLD-FET subtypes have been proposed, neuronal intermediate filament inclusion body disease (NIFID) and basophilic inclusion body disease (BIBD) 5,6 , which have more heterogenous clinical presentations including FTD, motor neuron diseases (MND) and movement disorders. Here, we used electron cryo-microscopy (cryo-EM) to determine a total of 32 amyloid filament structures from the brains of 17 individuals encompassing all three proposed subtypes of FTLD-FET and their diverse clinical presentations. All cases were characterised by TAF15 filaments, in the absence of filaments of the other FET proteins, fused in sarcoma (FUS) and Ewing's sarcoma (EWS). All three aFTLD-U cases had the previously-reported TAF15 fold 3 . Unexpectedly, we found four distinct TAF15 folds among 11 NIFID cases. Eight of these cases shared a common fold, while the remaining three were each distinct. Furthermore, we found distinct TAF15 folds for each of the three BIBD cases. Neuropathological reassessment of the neocortical TAF15 inclusion pathology of these cases distinguished the NIFID cases with the common fold from the others. Thus, TAF15 filament structures form the basis of a new, expanded classification of FTLD-FET subtypes. Moreover, we discovered a TAF15 Y38C variant in the filament fold of one of the individuals with BIBD. The structure is unable to incorporate wild-type TAF15, despite the individual being heterozygous, suggesting that this variant drives TAF15 filament assembly. This study provides structural and genetic evidence that TAF15 amyloid filaments underlie the diverse group of neurodegenerative diseases currently termed FTLD-FET, which we therefore rename FTLD-TAF15.
- MRC Laboratory of Molecular Biology, Cambridge, UK.
Organizational Affiliation: