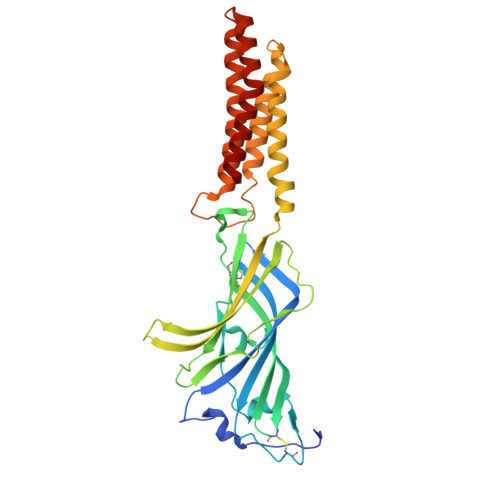
Structure of a pH-sensitive pentameric ligand-gated ion channel from the Sarcoptes scabies mite.
Kleiz-Ferreira, J., Brams, M., Harrison, P.J., Gallagher, C.I., Nys, M., Donze, Y., Quigley, A., Bertrand, D., Ulens, C.(2026) Nat Commun 17
- PubMed: 41820392 Search on PubMed
- DOI: https://doi.org/10.1038/s41467-026-70575-0
- Primary Citation Related Structures:
9RGM, 9RGN, 9RGO, 9RGP - PubMed Abstract:
Scabies is a skin infestation caused by the mite Sarcoptes scabiei and represents a substantial global health burden exacerbated by emerging resistance to ivermectin. An anionic pentameric ligand-gated ion channel from the mite, SsCl, shows pH-sensitivity and is significantly modulated by ivermectin. Here, we use cryo-EM and electrophysiology to explore the pH-sensing mechanisms of SsCl and the impact of ivermectin on channel activity. Structures of SsCl were resolved in closed (pH 6.5) and desensitized (pH 9) states, alongside ivermectin-bound conformations. The desensitized structure adopts an unexpected hourglass conformation, suggesting a gating mechanism closer related to cation-selective channels. Structural analysis and mutagenesis identify extracellular histidine and glutamic acid residues that impact the pH-sensitivity, likely contributing to a broader pH-sensing network. Ivermectin-bound structures reveal pH-dependent modulation, enhancing open-state prevalence at pH 9 and enabling atypical activation at pH 6.5. These findings offer initial insights into SsCl's pH-sensitivity and ivermectin's activity, informing next-generation antiparasitic design.
- Laboratory of Structural Neurobiology, Department of Cellular and Molecular Medicine, Faculty of Medicine, KU Leuven, Leuven, Belgium. jessica.kleiz@kuleuven.be.
Organizational Affiliation: