CPS secretion pathway Wza-Wzc (Conf 5)
Yuan, B., Heinz, D.W.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative polysaccharide export protein Wza | 394 | Escherichia coli K-12 | Mutation(s): 0 Gene Names: wza, b2062, JW2047 | 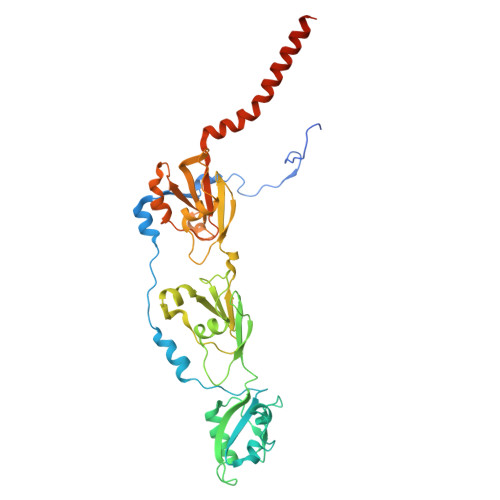 | |
UniProt | |||||
Find proteins for P0A930 (Escherichia coli (strain K12)) Explore P0A930 Go to UniProtKB: P0A930 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0A930 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Tyrosine-protein kinase wzc | 738 | Escherichia coli K-12 | Mutation(s): 1 Gene Names: wzc, b2060, JW2045 EC: 2.7.10 | 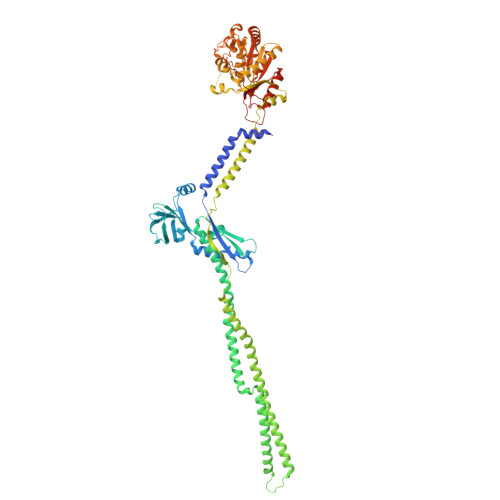 | |
UniProt | |||||
Find proteins for P76387 (Escherichia coli (strain K12)) Explore P76387 Go to UniProtKB: P76387 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P76387 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ADP (Subject of Investigation/LOI) Query on ADP | AA [auth K] BA [auth L] CA [auth M] DA [auth N] EA [auth O] | ADENOSINE-5'-DIPHOSPHATE C10 H15 N5 O10 P2 XTWYTFMLZFPYCI-KQYNXXCUSA-N |  | ||
| Funding Organization | Location | Grant Number |
|---|---|---|
| Helmholtz Association | Germany | -- |