Tumor suppressors BCDX2-CX3 supercomplex and DX2-CX3 complex template RAD51 filament formation
Koo, C.W., Ciferri, C., Yatskevich, S.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein RAD51 homolog 3 | A [auth C] | 376 | Homo sapiens | Mutation(s): 0 Gene Names: RAD51C, RAD51L2 | 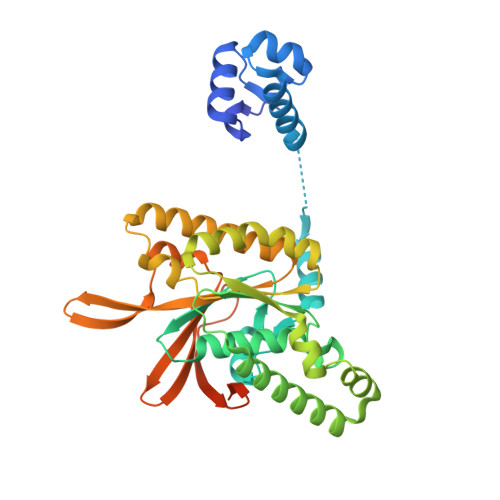 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O43502 GTEx: ENSG00000108384 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O43502 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein RAD51 homolog 4 | B [auth D] | 338 | Homo sapiens | Mutation(s): 0 Gene Names: RAD51D, RAD51L3 | 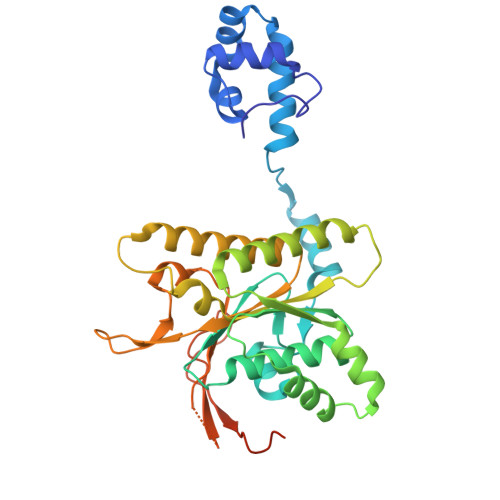 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O75771 GTEx: ENSG00000185379 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O75771 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein RAD51 homolog 1 | C [auth G], E [auth I], F [auth J], G [auth K], H [auth L] | 339 | Homo sapiens | Mutation(s): 0 Gene Names: RAD51, RAD51A, RECA EC: 3.6.4 | 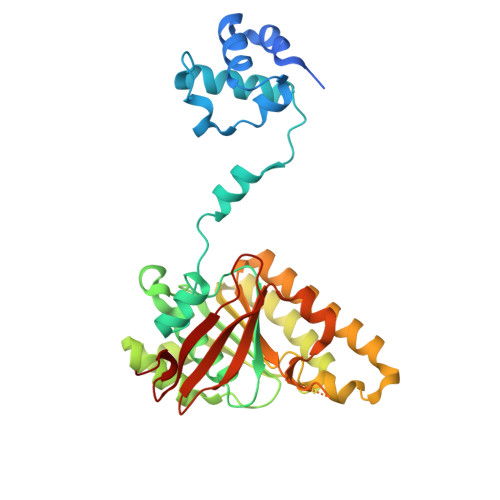 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q06609 GTEx: ENSG00000051180 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q06609 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein XRCC3 | I [auth R] | 346 | Homo sapiens | Mutation(s): 0 Gene Names: XRCC3 | 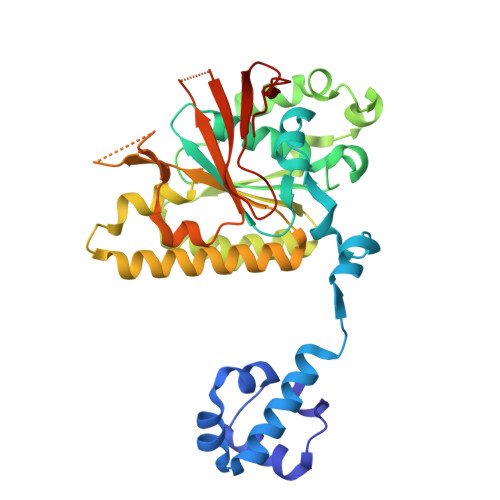 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O43542 GTEx: ENSG00000126215 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O43542 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA repair protein XRCC2 | J [auth X] | 280 | Homo sapiens | Mutation(s): 0 Gene Names: XRCC2 |  |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O43543 GTEx: ENSG00000196584 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O43543 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | ||||
| Molecule | Chains | Length | Organism | Image |
|---|---|---|---|---|
| DNA (26-MER) | D [auth H] | 30 | synthetic construct |  |
Sequence AnnotationsExpand | ||||
Reference Sequence | ||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ATP Download:Ideal Coordinates CCD File | CA [auth R] M [auth D] N [auth D] P [auth G] U [auth J] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| ADP Download:Ideal Coordinates CCD File | K [auth C] | ADENOSINE-5'-DIPHOSPHATE C10 H15 N5 O10 P2 XTWYTFMLZFPYCI-KQYNXXCUSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | AA [auth K] BA [auth L] DA [auth R] EA [auth X] L [auth C] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | cryoSPARC |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Not funded | -- |