Structure and Mechanism of the Prenylated-FMN Maturase, PhdC
DiRocco, D.J., Kilde, I., Langford, D.P., Roy, P., Bhaumik, S., Mendoza, J., Koutmos, M., Marsh, E.N.G.(2026) ACS Catal 16: 1773-1782
Experimental Data Snapshot
Starting Model: experimental
View more details
(2026) ACS Catal 16: 1773-1782
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Pyridoxamine 5'-phosphate oxidase putative domain-containing protein | 168 | Mycolicibacterium fortuitum | Mutation(s): 0 Gene Names: XA26_16660 | 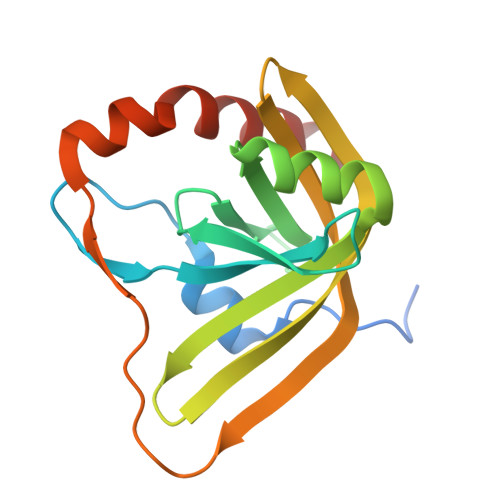 | |
UniProt | |||||
Find proteins for A0A0N9XAG5 (Mycolicibacterium fortuitum) Explore A0A0N9XAG5 Go to UniProtKB: A0A0N9XAG5 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0N9XAG5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 4LU (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A], G [auth B], I [auth C], K [auth D] | 1-deoxy-5-O-phosphono-1-(3,3,4,5-tetramethyl-9,11-dioxo-2,3,8,9,10,11-hexahydro-7H-quinolino[1,8-fg]pteridin-12-ium-7-y
l)-D-ribitol C22 H30 N4 O9 P KOUJZPGFPGLHCZ-IYOUNJFTSA-O |  | ||
| NA Download:Ideal Coordinates CCD File | F [auth A], H [auth B], J [auth C], L [auth D] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 53.552 | α = 69.95 |
| b = 60.19 | β = 85.032 |
| c = 65.053 | γ = 65.414 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| xia2 | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| REFMAC | refinement |
| Coot | model building |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Science Foundation (NSF, United States) | United States | CHE 2203729 |
| National Science Foundation (NSF, United States) | United States | CHE 1904759 |