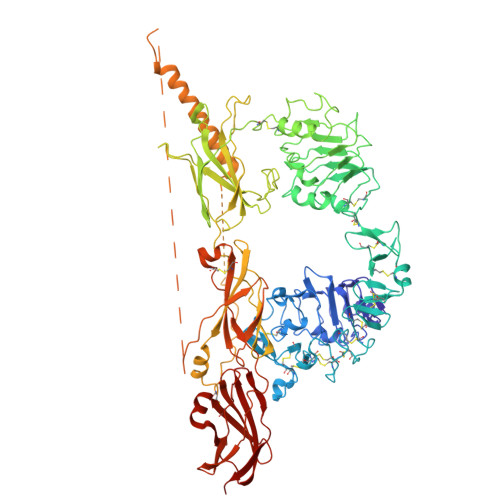
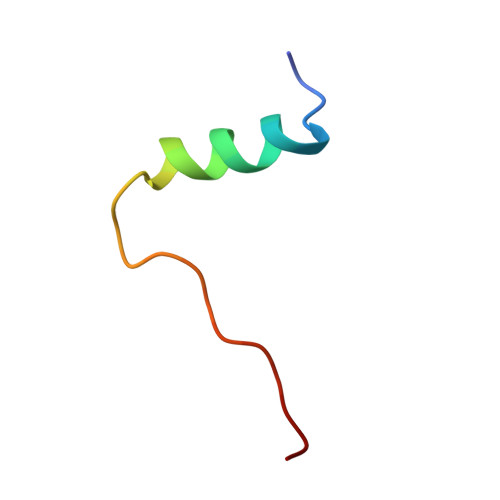
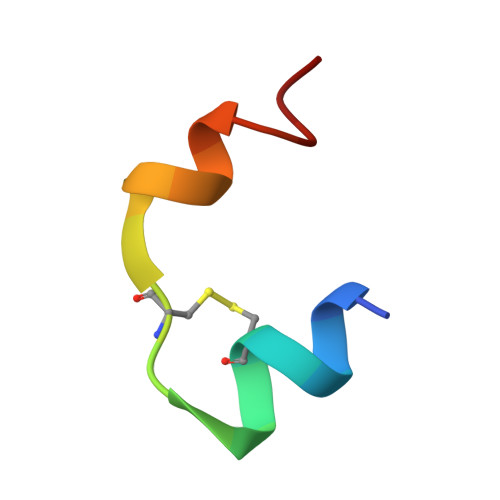
Neuroprotective potential of an engineered hormone with dual insulin and IGF-1 receptor activity
Selicharova, I., Kirk, N.S., Lubos, M., Kertisova, A., Mitrova, K., Zakova, L., Chrudinova, M., Brezinova, J., Harant, K., Voldrich, J., Panikova, T., Torres-Aleman, I., Huffman, D., Jiracek, J.To be published.