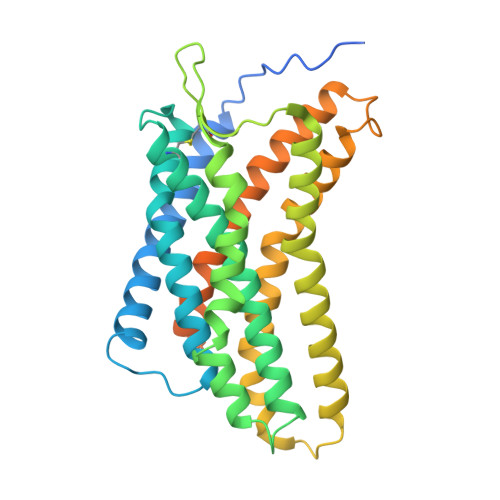
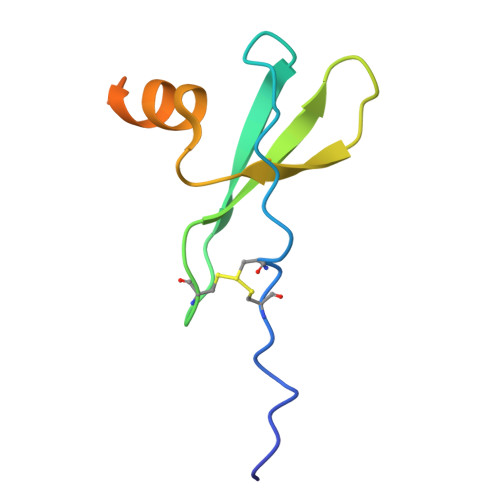
CXCR4 mediated recognition of HIV envelope spike and inhibition by CXCL12.
Zhang, Z., Zhang, H., Zheng, L., Chen, S., Du, S., Xiao, J., Patel, D.J.(2025) Nat Commun 16: 8653-8653
- PubMed: 41027939 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-63815-2
- Primary Citation Related Structures:
9ME1, 9MEJ, 9MEN, 9MET, 9MEU - PubMed Abstract:
CCR5 and CXCR4 both act as HIV co-receptors, though CXCR4 is less explored. CXCR4 binds the chemokine CXCL12 to regulate cellular processes and mediate HIV entry, a process that CXCL12 inhibits. Using cryo-EM, we investigate HIV-2 envelope (Env) spike recognition by CXCR4 and how CXCL12 inhibit this interaction. We discover that CXCR4 unexpected forms a tetramer, both alone and in complex. It binds CXCL12 with 4:8 and 8:8 stoichiometries, with the CXCL12 N-terminus inserting into the CXCR4 pocket. Structures of CXCR4-gp120 HIV-2 complex show one or two gp120 molecules per CXCR4 tetramer, with the V3 loop occupying the major sub-pocket of CXCR4 through deep embedment of its GFKF motif. The CXCL12 N-terminus chashes with gp120 HIV-2 V3 loops, explain its inhibitory effect. Docking analyses of other HIV antagonists further clarify their mechanisms. The CXCR4-gp120 HIV-1 model illustrate how V3 loop residues define co-receptor specificity, offering insights into co-receptor switching and therapeutic design.
- Structural Biology Program, Memorial Sloan-Kettering Cancer Center, New York, NY, USA. zhangz7@mskcc.org.
Organizational Affiliation: