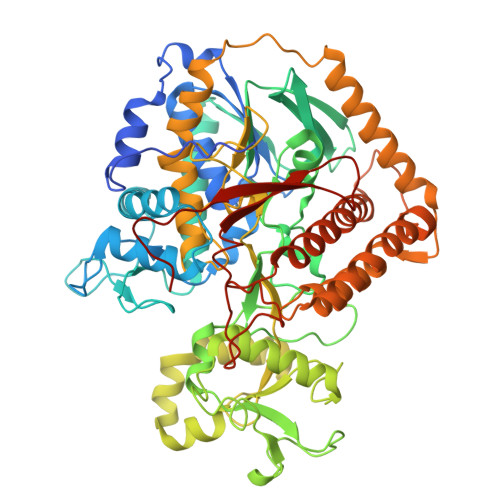
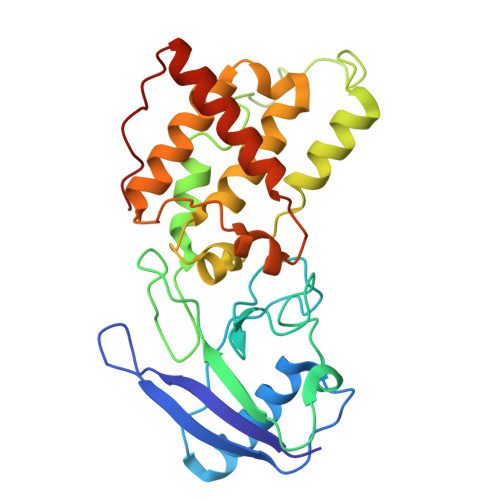
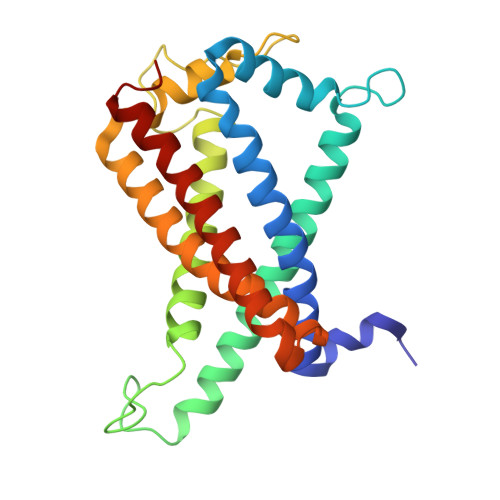
Structural basis of menaquinone reduction by succinate dehydrogenase from Chloroflexus aurantiacus.
Zhang, X., Wu, J., Wang, J., He, H., Liu, A., Hong, X., Yu, Y., Pei, X., Fang, X., Xin, Y., Yu, L., Tian, C., Xu, X.(2025) Nat Commun 16: 10782-10782
- PubMed: 41315263 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-65828-3
- Primary Citation Related Structures:
9LAY, 9LAZ, 9LB0, 9LB1 - PubMed Abstract:
Succinate: menaquinone oxidoreductase (SQR) couples the oxidation of succinate with the reduction of menaquinone (MK) as part of the TCA cycle and the aerobic respiratory chain in MK-containing bacteria and archaea. Despite its significance, questions persist regarding the electron and proton transfer mechanisms that drive the endergonic MK reduction by succinate. In this study, we determine cryo-EM structures of succinate dehydrogenase (SDH) from Chloroflexus aurantiacus (CaSDH), a facultative filamentous anoxygenic phototroph (FAP) that forms one of the earliest branches of photosynthetic bacteria. The structures of trimeric CaSDH, resolved in both apo- and MK-bound forms, reveal a single membrane-anchoring subunit containing two b-type hemes, a canonical Q P site, and a Q D site with atypical location, configuration and specificity, each bound to MK molecules. Using structural analysis, EPR, and enzymatic assays, we uncover electron transfer pathways connecting succinate oxidation to MK reduction at the Q P and Q D sites. These findings provide structural insights into the electron and proton transfer mechanisms of MK-dependent diheme SQRs and establish a foundation for structure-based inhibitor design and antibacterial drug development targeting these enzymes.
- Zhejiang Key Laboratory of Medical Epigenetics, Hangzhou Normal University, Hangzhou, China.
Organizational Affiliation: