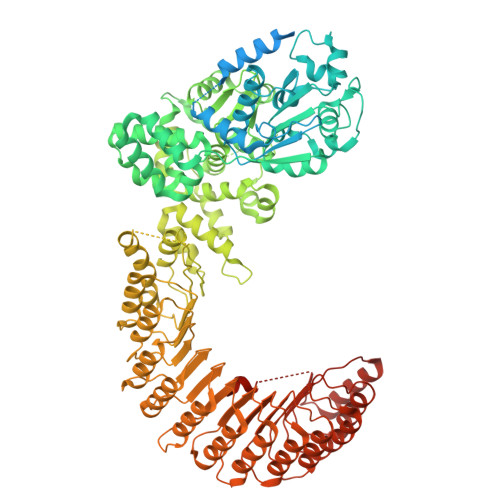
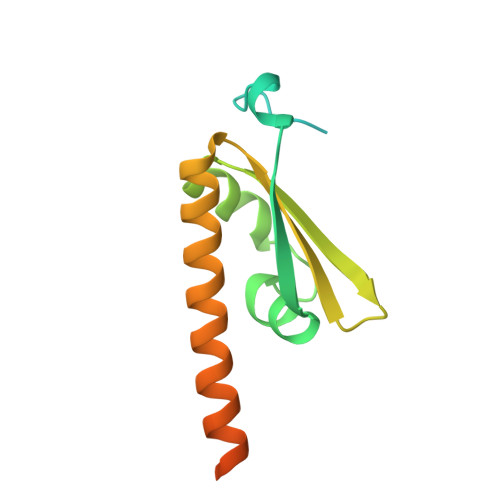
Structural assembly of the subcortical maternal complex SCMC.
Ou, G., Liu, Q., Jiao, H., Han, Z., Li, J., Min, L., Chi, P., Liu, S., Li, J., Qi, Q., Zhang, Z., Guo, L., Wang, X., Li, L., Chen, J., Hu, H., Deng, D.(2026) Structure 34: 20-31.e7
- PubMed: 41118754 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2025.09.009
- Primary Citation Related Structures:
9L4J, 9L4K, 9L4L - PubMed Abstract:
The subcortical maternal complex (SCMC) is essential for mammalian preimplantation development, yet how SCMC core (MATER/NLRP5, TLE6, FLOPED/OOEP) engages regulatory partners remains unclear. We determined cryo-EM structures of mouse SCMC bound to ZBED3 and human SCMC bound to NLRP2. Our structure reveals that ZBED3 interacts with all three SCMC core subunits via its zinc finger domain, with conserved residue Phe73 mediating specific contacts. In contrast, human NLRP2 only binds to the WD40 domain of TLE6 through its leucine-rich repeat (LRR) domain. Similar interactions were also confirmed for NLRP7 with TLE6. These findings were cross-validated by in vivo proximity ligation and in vitro pull-down assays. Our work proposes a paradigmatic "Lego-like" assembly model, where the SCMC core sequentially recruits different partners through diverse molecular interfaces. These findings provide critical structural insights into the SCMC's architecture and its multifaceted regulatory roles in early mammalian embryogenesis.
- Department of Laboratory Medicine, Key Laboratory of Birth Defects and Related Disease of Women and Children of MOE, State Key Laboratory of Biotherapy, West China Second Hospital, Sichuan University, Chengdu 610041, China; Development and Related Diseases of Women and Children Key Laboratory of Sichuan Province, Sichuan University, Chengdu 610041, China.
Organizational Affiliation: