Purification and Electron Transfer from Soluble c-Type Cytochrome TorC to TorA for Trimethylamine N-Oxide Reduction.
Panwar, A., Martins, B.M., Sommer, F., Schroda, M., Dobbek, H., Iobbi-Nivol, C., Jourlin-Castelli, C., Leimkuhler, S.(2024) Int J Mol Sci 25
- PubMed: 39769096 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/ijms252413331
- Primary Citation Related Structures:
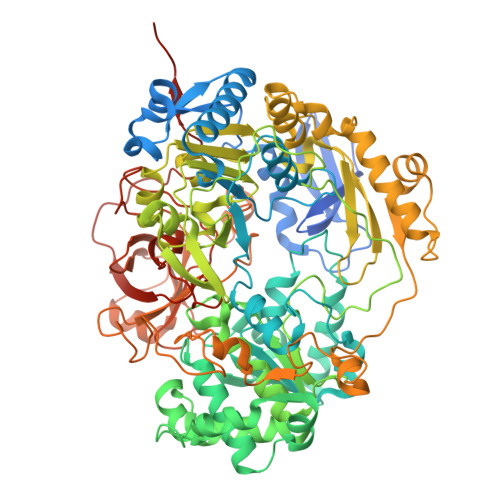
9H4T - PubMed Abstract:
The enterobacterium Escherichia coli present in the human gut can reduce trimethylamine N-oxide (TMAO) to trimethylamine during anaerobic respiration. The TMAO reductase TorA is a monomeric, bis-molybdopterin guanine dinucleotide (bis-MGD) cofactor-containing enzyme that belongs to the dimethyl sulfoxide reductase family of molybdoenzymes. TorA is anchored to the membrane via TorC, a pentahemic c -type cytochrome which receives the electrons from the menaquinol pool. Here, we designed an expression system for the production of a stable soluble form of multiheme-containing TorC, providing, for the first time, the purification of a soluble pentahemic cytochrome- c from E. coli . Our focus was to investigate the interaction between TorA and soluble TorC to establish the electron transfer pathway. We solved the X-ray structure of E. coli TorA and performed chemical crosslinking of TorA and TorC. Another goal was to establish an activity assay that used the physiological electron transfer pathway instead of the commonly used unphysiological electron donors methylviologen or benzylviologen. An AlphaFold model including the crosslinking sites provided insights into the electron transfer between TorC C and the active site of TorA.
- Department of Molecular Enzymology, Institute of Biochemistry and Biology, University of Potsdam, Karl-Liebknecht Str. 24-25, 14476 Potsdam, Germany.
Organizational Affiliation: