Minimal supercomplexes in alphaproteobacteria reveal conserved structural mechanisms for efficient respiration
Yaikhomba, M., Hirst, J., Croll, T.I., Spikes, T.E.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit A | A, B [auth A1] | 121 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 | 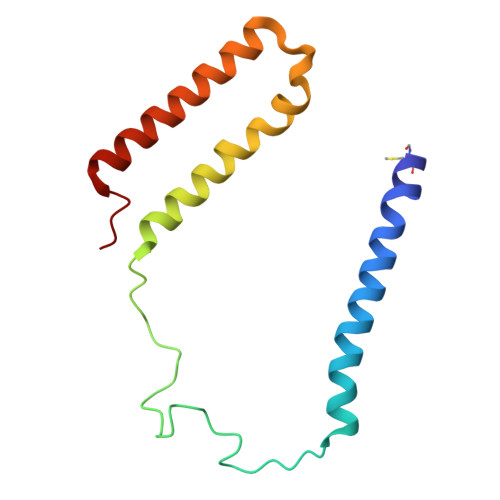 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B498 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit B | C [auth B], D [auth B1] | 175 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 | 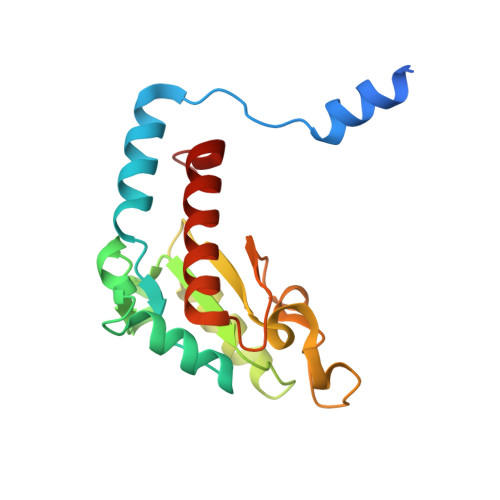 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B497 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit C | E [auth C], F [auth C1] | 208 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 | 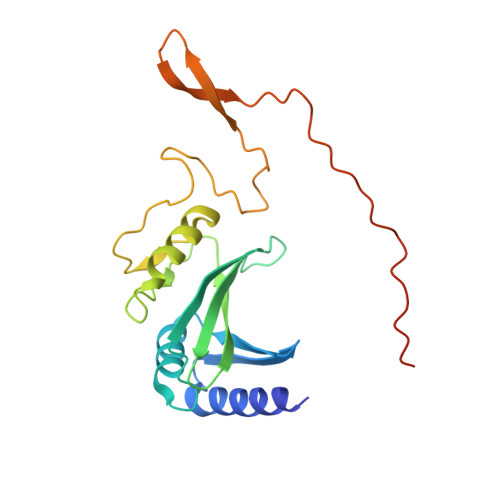 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B496 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit D | G [auth D], H [auth D1] | 412 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 | 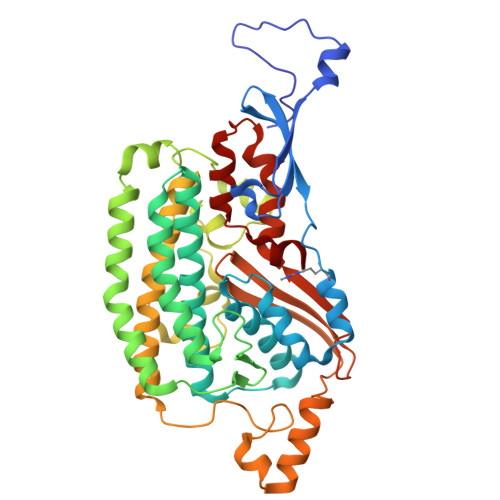 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B495 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit E | I [auth E], J [auth E1] | 239 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B494 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit F | K [auth F], L [auth F1] | 431 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B491 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 7 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase | M [auth G], N [auth G1] | 674 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B489 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 8 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit H | O [auth H], P [auth H1] | 345 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B487 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 9 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit I | Q [auth I], R [auth I1] | 163 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B486 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 10 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit J | S [auth J], T [auth J1] | 199 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B483 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 11 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit K | U [auth K], V [auth K1] | 101 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B482 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 12 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit L | W [auth L], X [auth L1] | 703 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B481 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 13 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH dehydrogenase subunit M | Y [auth M], Z [auth M1] | 513 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 1.6.5.3 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B480 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 14 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH-quinone oxidoreductase subunit N | AA [auth N], BA [auth N1] | 499 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B479 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 15 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NAD-dependent epimerase/dehydratase | CA [auth P], DA [auth P1] | 330 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1AZB0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 16 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ETC complex I subunit conserved region | EA [auth Q], FA [auth Q1] | 103 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B1M0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 17 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Zinc finger CHCC-type domain-containing protein | GA [auth R], HA [auth R1] | 62 | Paracoccus denitrificans PD1222 | Mutation(s): 0 | 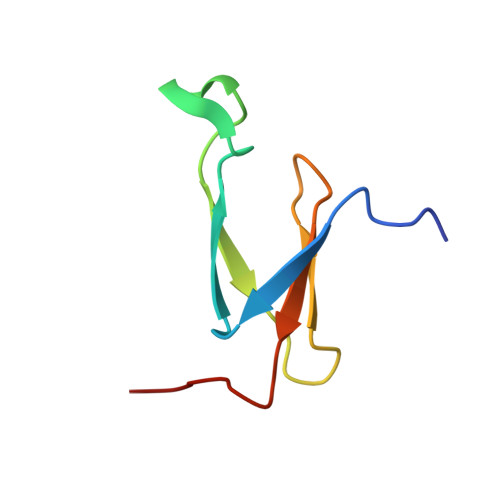 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B357 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 18 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein-L-isoaspartate O-methyltransferase | IA [auth Z], JA [auth Z1] | 217 | Paracoccus denitrificans PD1222 | Mutation(s): 0 | 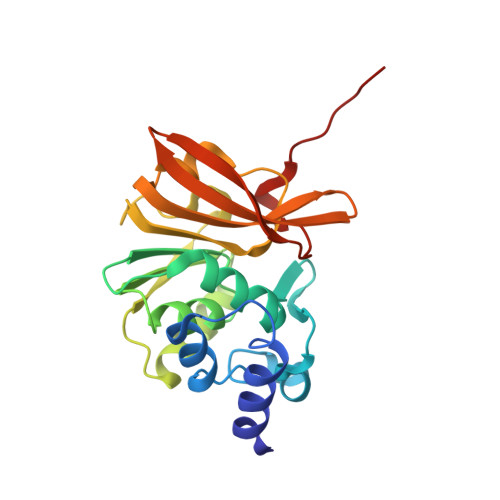 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B5L6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 19 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome b | KA [auth a], NA [auth d] | 440 | Paracoccus denitrificans PD1222 | Mutation(s): 0 | 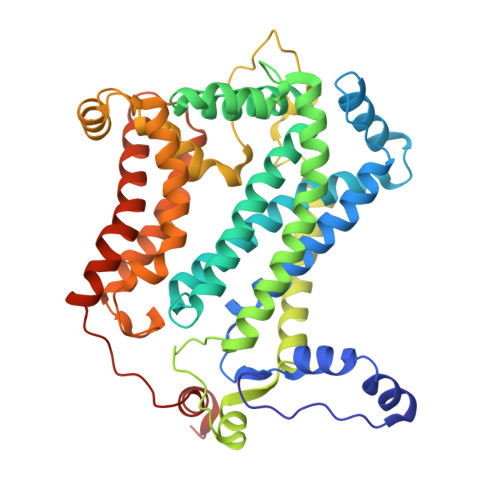 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 20 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c1 | LA [auth b], OA [auth e] | 450 | Paracoccus denitrificans PD1222 | Mutation(s): 0 | 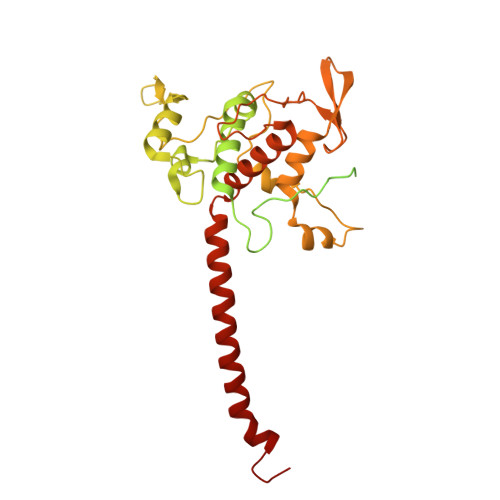 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 21 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Ubiquinol-cytochrome c reductase iron-sulfur subunit | MA [auth c], PA [auth f] | 195 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.8 | 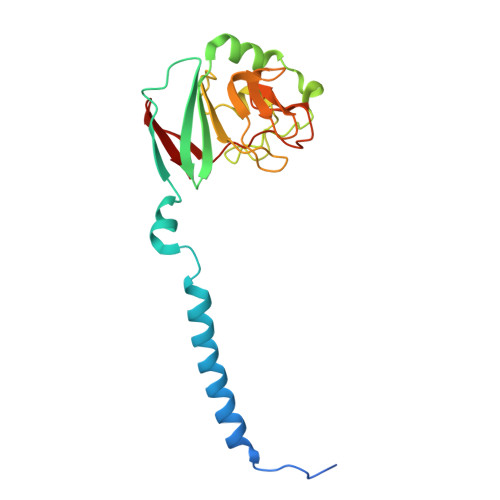 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B4F2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 22 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c oxidase subunit 1 | QA [auth g], UA [auth k] | 558 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 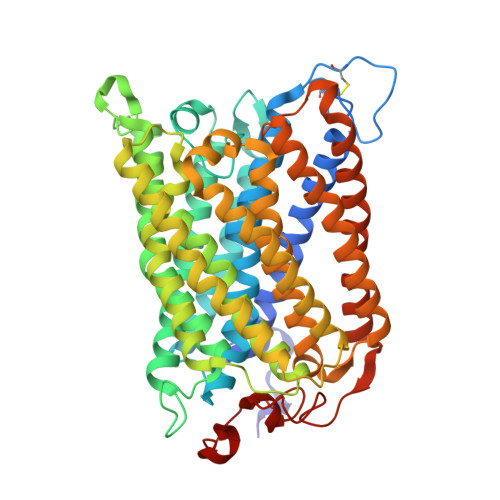 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B3D7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 23 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c oxidase subunit 2 | RA [auth h], VA [auth l] | 298 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 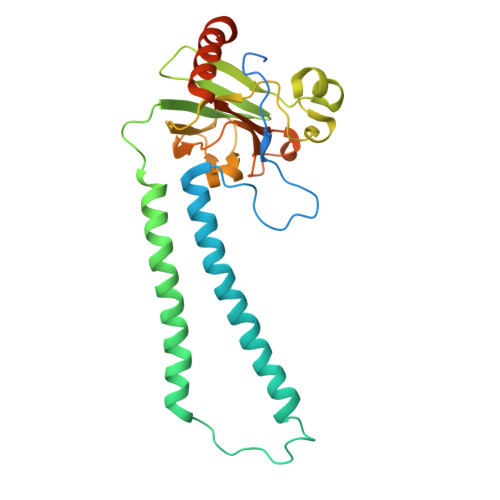 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1BA41 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 24 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| cytochrome-c oxidase | SA [auth i], WA [auth m] | 274 | Paracoccus denitrificans PD1222 | Mutation(s): 0 EC: 7.1.1.9 | 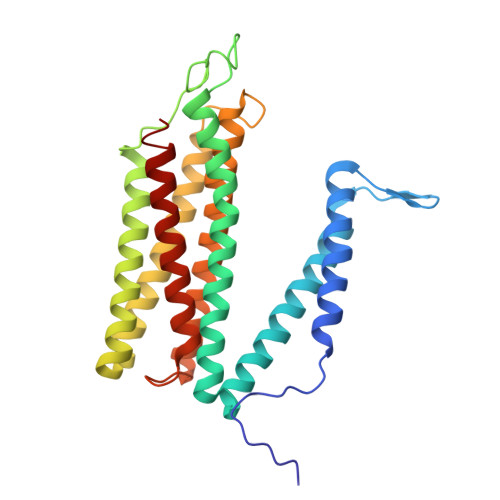 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1BA37 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 25 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Aa3 type cytochrome c oxidase subunit IV | TA [auth j], XA [auth n] | 66 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1AZ52 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 26 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome c, class I | YA [auth o], ZA [auth p] | 176 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B311 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 27 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| NADH:ubiquinone oxidoreductase 17.2 kD subunit | AB [auth q], BB [auth q1] | 124 | Paracoccus denitrificans PD1222 | Mutation(s): 0 |  |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A1B1H8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 18 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CDL Download:Ideal Coordinates CCD File | UC [auth a] | CARDIOLIPIN C81 H156 O17 P2 XVTUQDWPJJBEHJ-KZCWQMDCSA-L |  | ||
| U10 Download:Ideal Coordinates CCD File | CD [auth d] DB [auth B] FB [auth B1] QC [auth a] SC [auth a] | UBIQUINONE-10 C59 H90 O4 ACTIUHUUMQJHFO-UPTCCGCDSA-N |  | ||
| HEA (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AE [auth k], JD [auth g], ND [auth g], YD [auth k] | HEME-A C49 H56 Fe N4 O6 ZGGYGTCPXNDTRV-PRYGPKJJSA-L |  | ||
| P5S Download:Ideal Coordinates CCD File | EC [auth L], HC [auth L1] | O-[(R)-{[(2R)-2,3-bis(octadecanoyloxy)propyl]oxy}(hydroxy)phosphoryl]-L-serine C42 H82 N O10 P TZCPCKNHXULUIY-RGULYWFUSA-N |  | ||
| PC1 Download:Ideal Coordinates CCD File | FC [auth L1] FE [auth m] GE [auth m] JC [auth M] JE [auth n] | 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE C44 H88 N O8 P NRJAVPSFFCBXDT-HUESYALOSA-N |  | ||
| 3PE Download:Ideal Coordinates CCD File | CC [auth L] CE [auth k] ED [auth e] EE [auth m] GC [auth L1] | 1,2-Distearoyl-sn-glycerophosphoethanolamine C41 H82 N O8 P LVNGJLRDBYCPGB-LDLOPFEMSA-N |  | ||
| 3PH Download:Ideal Coordinates CCD File | DC [auth L] HD [auth f] IC [auth L1] KD [auth g] KE [auth p] | 1,2-DIACYL-GLYCEROL-3-SN-PHOSPHATE C39 H77 O8 P YFWHNAWEOZTIPI-DIPNUNPCSA-N |  | ||
| HEC (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | GD [auth e], XC [auth b] | HEME C C34 H34 Fe N4 O4 HXQIYSLZKNYNMH-LJNAALQVSA-N |  | ||
| HEM (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | AD [auth d], DD [auth d], PC [auth a], RC [auth a] | PROTOPORPHYRIN IX CONTAINING FE C34 H32 Fe N4 O4 KABFMIBPWCXCRK-RGGAHWMASA-L |  | ||
| DU0 Download:Ideal Coordinates CCD File | BC [auth K], BD [auth d], KC [auth M], MC [auth M1], TC [auth a] | 2-[2-[(1~{S},2~{S},4~{S},5'~{R},6~{R},7~{S},8~{R},9~{S},12~{S},13~{R},16~{S})-5',7,9,13-tetramethylspiro[5-oxapentacyclo[10.8.0.0^{2,9}.0^{4,8}.0^{13,18}]icos-18-ene-6,2'-oxane]-16-yl]oxyethyl]propane-1,3-diol C32 H52 O5 GFDJQXOBWHMOSQ-LEZUHYJESA-N |  | ||
| FMN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | LB [auth F], MB [auth F1] | FLAVIN MONONUCLEOTIDE C17 H21 N4 O9 P FVTCRASFADXXNN-SCRDCRAPSA-N |  | ||
| SF4 Download:Ideal Coordinates CCD File | AC [auth I1] CB [auth B] EB [auth B1] KB [auth F] NB [auth F1] | IRON/SULFUR CLUSTER Fe4 S4 LJBDFODJNLIPKO-UHFFFAOYSA-N |  | ||
| FES Download:Ideal Coordinates CCD File | IB [auth E] ID [auth f] JB [auth E1] QB [auth G] RB [auth G1] | FE2/S2 (INORGANIC) CLUSTER Fe2 S2 NIXDOXVAJZFRNF-UHFFFAOYSA-N |  | ||
| CUA Download:Ideal Coordinates CCD File | DE [auth l], QD [auth h] | DINUCLEAR COPPER ION Cu2 ALKZAGKDWUSJED-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | IE [auth m], NC [auth R], OC [auth R1], SD [auth i] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CU Download:Ideal Coordinates CCD File | LD [auth g], ZD [auth k] | COPPER (II) ION Cu JPVYNHNXODAKFH-UHFFFAOYSA-N |  | ||
| MN Download:Ideal Coordinates CCD File | BE [auth k], OD [auth g] | MANGANESE (II) ION Mn WAEMQWOKJMHJLA-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | FD [auth e] GB [auth D] HB [auth D1] MD [auth g] VC [auth b] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| FME Query on FME | A, B [auth A1] | L-PEPTIDE LINKING | C6 H11 N O3 S |  | MET |
| 2MR Query on 2MR | G [auth D], H [auth D1] | L-PEPTIDE LINKING | C8 H18 N4 O2 |  | ARG |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MC_UU_00015/2 and MC_UU_00028/1 |