The HIV protease inhibitor amprenavir binding to the active site of Cryphonectria parasitica endothiapepsin
Falke, S., Senst, J.M., Guenther, S., Meents, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Endothiapepsin | 330 | Cryphonectria parasitica | Mutation(s): 0 Gene Names: EAPA, EPN-1 EC: 3.4.23.22 | 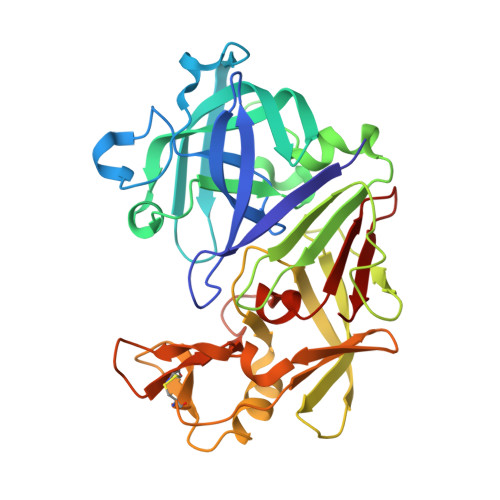 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P11838 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 478 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A] | {3-[(4-AMINO-BENZENESULFONYL)-ISOBUTYL-AMINO]-1-BENZYL-2-HYDROXY-PROPYL}-CARBAMIC ACID TETRAHYDRO-FURAN-3-YL ESTER C25 H35 N3 O6 S YMARZQAQMVYCKC-OEMFJLHTSA-N |  | ||
| PG4 Download:Ideal Coordinates CCD File | C [auth A], D [auth A], I [auth A] | TETRAETHYLENE GLYCOL C8 H18 O5 UWHCKJMYHZGTIT-UHFFFAOYSA-N |  | ||
| PGE Download:Ideal Coordinates CCD File | E [auth A], F [auth A] | TRIETHYLENE GLYCOL C6 H14 O4 ZIBGPFATKBEMQZ-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | G [auth A], K [auth A], L [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| ACY Download:Ideal Coordinates CCD File | H [auth A], J [auth A] | ACETIC ACID C2 H4 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 45.638 | α = 90 |
| b = 72.832 | β = 109.46 |
| c = 53.23 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Helmholtz Association | Germany | -- |
| German Federal Ministry for Education and Research | Germany | -- |