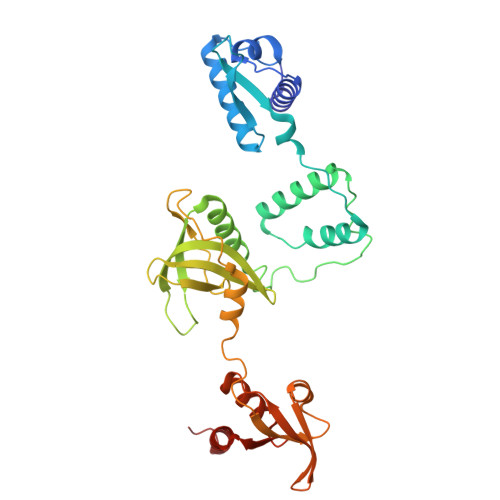
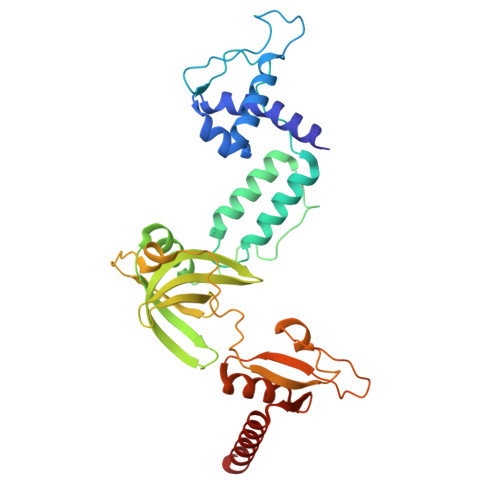
Single-stranded DNA binding to the transcription factor PafBC triggers the mycobacterial DNA damage response.
Schilling, C.M., Zdanowicz, R., Rabl, J., Muller, A.U., Boehringer, D., Glockshuber, R., Weber-Ban, E.(2025) Sci Adv 11: eadq9054-eadq9054
- PubMed: 39919186 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.adq9054
- Primary Citation Related Structures:
9FND, 9FNE - PubMed Abstract:
The DNA damage response in mycobacteria is controlled by the heterodimeric transcription factor PafBC, a member of the WYL domain-containing protein family. It has been shown that PafBC induces transcription of its regulon by reprogramming the housekeeping RNA polymerase holoenzyme to recognize PafBC-dependent promoters through sigma adaptation. However, the mechanism by which DNA damage is sensed and translated into PafBC activation has remained unclear. Here, we demonstrate that the binding of single-stranded DNA (ssDNA) to the WYL domains of PafBC activates the transcription factor. Our cryo-electron microscopy structure of full-length PafBC in its active conformation, bound to the transcription initiation complex, reveals a previously unknown mode of interaction between the ssDNA and the WYL domains. Using biochemical experiments, we show that short ssDNA fragments bind to PafBC dynamically, resulting in deactivation as ssDNA levels decrease postrepair. Our findings shed light on the mechanism linking DNA damage to PafBC activation and expand our understanding of WYL domain-containing proteins.
- ETH Zurich, Institute of Molecular Biology and Biophysics, 8093 Zurich, Switzerland.
Organizational Affiliation: