Ternary complex DNA-encoded library screening uncovers FKBP molecular glues
Zandi, T.A., Tan, Z.R., Romanowski, M.J., Viscomi, J.S., Tong, B.Q., Bonazzi, S., Zecri, F.J., Schreiber, S.L., Michaud, G.A.To be published.
Experimental Data Snapshot
Starting Models: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Peptidyl-prolyl cis-trans isomerase FKBP1A | 109 | Homo sapiens | Mutation(s): 0 EC: 5.2.1.8 | 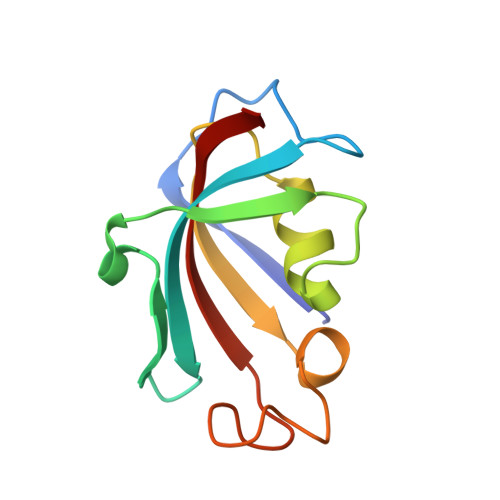 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P62942 GTEx: ENSG00000088832 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P62942 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Bromodomain-containing protein 9 | 116 | Homo sapiens | Mutation(s): 0 | 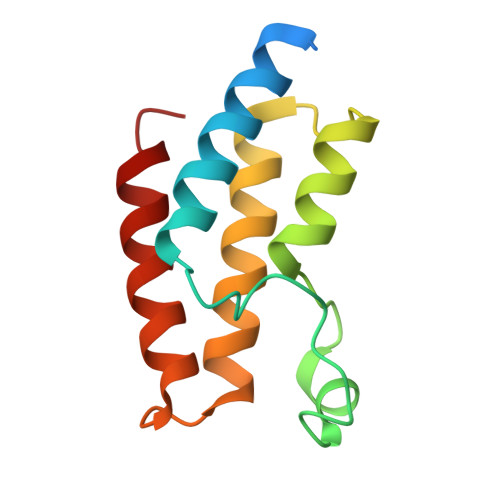 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9H8M2 GTEx: ENSG00000028310 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9H8M2 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| A1BB8 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth B], J [auth D], L [auth G], M [auth H] | 4-[4-{cyclopropyl[(1-methyl-1H-pyrazol-4-yl)methyl]amino}-6-({1-[(2R)-2-{[(2S)-1-(3,3-dimethyl-2-oxopentanoyl)piperidine-2-carbonyl]amino}-4-(4-methoxyphenyl)butanoyl]piperidin-4-yl}amino)-1,3,5-triazin-2-yl]-N-ethylpiperazine-1-carboxamide C47 H69 N13 O6 ADQZIARRQDOKQN-AMAPPZPBSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | K [auth F] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 59.163 | α = 90 |
| b = 97.512 | β = 93.69 |
| c = 100.002 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Other private | United States | -- |