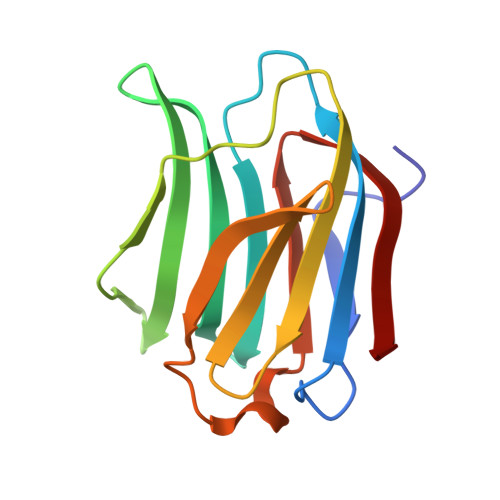
Exploring the interactive effect of pectins with galectin-3 to reduce the viability of T98G glioblastoma cell lineage
dos Santos, L.V., Dias, I.P., Mazepa, E., Minella, T.F., Baruffi, M.R., Mestriner, L., Nascimento, A.F.Z., Albuquerque, L.J.C., Mischiatti, K.L., Winnischofer, S.M.B., Amaral, S.S., Silveira, J.L.M., Picheth, G.F.To be published.