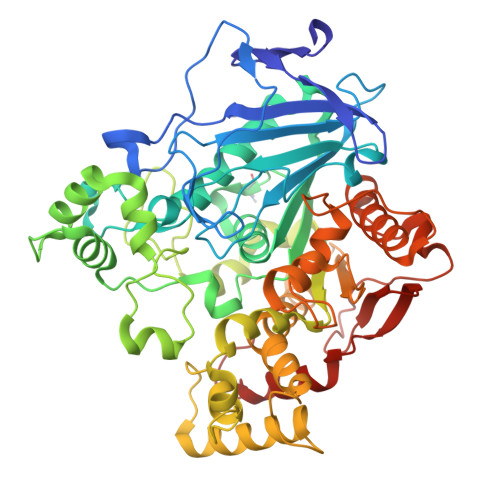
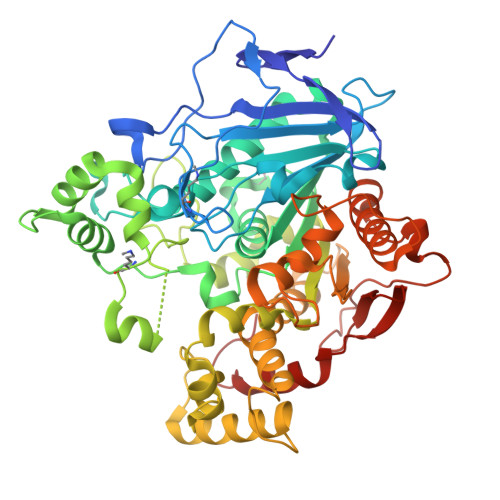
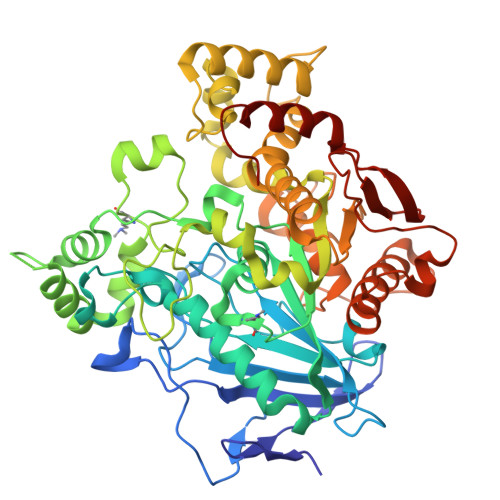
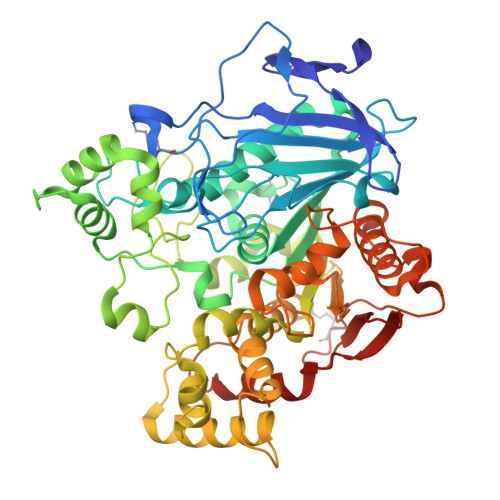
Structural and evolutionary constraints of organophosphate resistance in dipteran carboxylesterases.
Frkic, R.L., Giang, A., Pulsford, S.B., Liu, J.W., Esmaeily, M., Carr, P.D., Fraser, N.J., Hopkins, D., Oakeshott, J.G., Batterham, P., Mabbitt, P.D., Jackson, C.J.(2026) Proc Natl Acad Sci U S A 123: e2517957123-e2517957123
- PubMed: 41730108 Search on PubMed
- DOI: https://doi.org/10.1073/pnas.2517957123
- Primary Citation Related Structures:
9D1J, 9D1K, 9D1L, 9D1M, 9D1N, 9D1O, 9D1P, 9D1Q, 9D1R, 9D1S, 9D1T - PubMed Abstract:
Enzymatic detoxification of organophosphate (OP) insecticides can confer resistance in some insects, yet the precise molecular basis of this trait, and how it has evolved, remains poorly understood. In certain dipteran species, a G→D mutation in the oxyanion hole of α-carboxylesterases (CBEs) enhances OP hydrolysis, yet this adaptation is not widespread despite the presence of orthologous CBEs in other insect species that are also exposed to OPs. The extent, and molecular basis, of evolutionary contingency and epistasis in this catalytic OP resistance has not been explored, and how further mutations might optimize OP detoxification in the future is not clear. Here, we systematically compare OP hydrolysis and analyze structures of CBE orthologs across several dipteran species, revealing that the success of the G137D mutation is sequence context-dependent. We employed laboratory-directed evolution to enhance OP turnover over 1,000-fold vs. the wildtype enzyme and tested these variants in transgenic Drosophila melanogaster , demonstrating that improved catalytic rates do not directly translate to increased resistance. By highlighting the trade-off between organophosphate affinity and turnover, this work further clarifies the complex evolutionary trajectories determining why a particular resistance mechanism may evolve in some species but not others.
- Research School of Chemistry, Australian National University, Canberra, ACT 2601, Australia.
Organizational Affiliation: