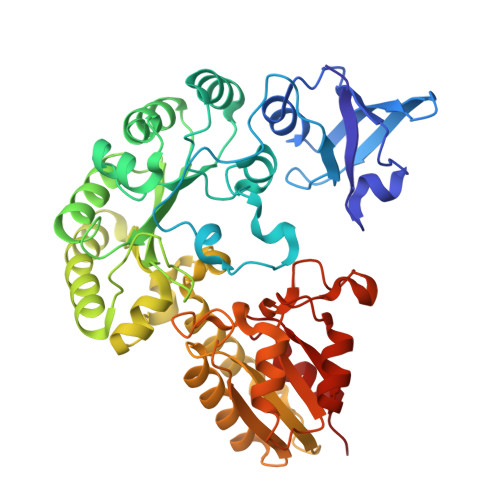
Structural Evidence for DUF512 as a Radical S -Adenosylmethionine Cobalamin-Binding Domain.
Wang, B., Solinski, A.E., Radle, M.I., Peduzzi, O.M., Knox, H.L., Cui, J., Maurya, R.K., Yennawar, N.H., Booker, S.J.(2024) ACS Bio Med Chem Au 4: 319-330
- PubMed: 39712206 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acsbiomedchemau.4c00067
- Primary Citation Related Structures:
9CG1, 9CG2 - PubMed Abstract:
Cobalamin (Cbl)-dependent radical S -adenosylmethionine (SAM) enzymes constitute a large subclass of radical SAM (RS) enzymes that use Cbl to catalyze various types of reactions, the most common of which are methylations. Most Cbl-dependent RS enzymes contain an N-terminal Rossmann fold that aids Cbl binding. Recently, it has been demonstrated that the methanogenesis marker protein 10 (Mmp10) requires Cbl to methylate an arginine residue in the α-subunit of methyl coenzyme M reductase. However, Mmp10 contains a Cbl-binding domain in the C-terminal region of its primary structure that does not share significant sequence similarity with canonical RS Cbl-binding domains. Bioinformatic analysis of Mmp10 identified DUF512 (Domain of Unknown Function 512) as a potential Cbl-binding domain in RS enzymes. In this paper, four randomly selected DUF512-containing proteins from various organisms were overexpressed, purified, and shown to bind Cbl. X-ray crystal structures of DUF512-containing proteins from Clostridium sporogenes and Pyrococcus furiosus were determined, confirming their C-terminal Cbl-binding domains. The structure of the DUF512-containing protein from C. sporogenes is the first of an RS enzyme containing a PDZ domain. Its RS domain has an unprecedented β 3 α 4 core, whereas most RS enzymes adopt a (βα) 6 core. The DUF512-containing protein from P. furiosus has no PDZ domain, but its RS domain also has an uncommon (βα) 5 core.
- Department of Chemistry, The Pennsylvania State University, University Park, Pennsylvania 16802, United States.
Organizational Affiliation: