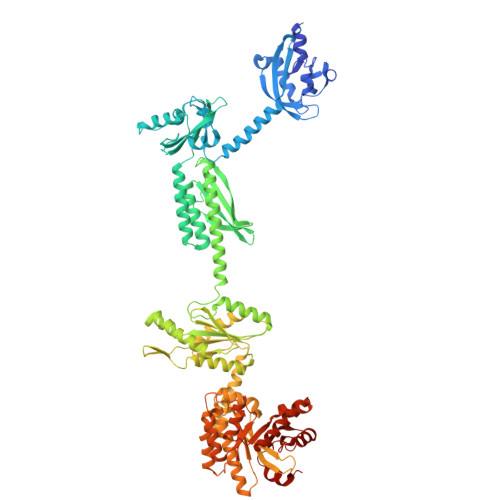
Structures of the multi-domain oxygen sensor DosP: remote control of a c-di-GMP phosphodiesterase by a regulatory PAS domain.
Wu, W., Kumar, P., Brautigam, C.A., Tso, S.C., Baniasadi, H.R., Kober, D.L., Gilles-Gonzalez, M.A.(2024) Nat Commun 15: 9653-9653
- PubMed: 39511182 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-024-53942-7
- Primary Citation Related Structures:
9BGV, 9BKV, 9CDR, 9CE0, 9CLO, 9CMF - PubMed Abstract:
The heme-based direct oxygen sensor DosP degrades c-di-GMP, a second messenger nearly unique to bacteria. In stationary phase Escherichia coli, DosP is the most abundant c-di-GMP phosphodiesterase. Ligation of O 2 to a heme-binding PAS domain (hPAS) of the protein enhances the phosphodiesterase through an allosteric mechanism that has remained elusive. We determine six structures of full-length DosP in its aerobic or anaerobic conformations, with or without c-di-GMP. DosP is an elongated dimer with the regulatory heme containing domain and phosphodiesterase separated by nearly 180 Å. In the absence of substrate, regardless of the heme status, DosP presents an equilibrium of two distinct conformations. Binding of substrate induces DosP to adopt a single, ON-state or OFF-state conformation depending on its heme status. Structural and biochemical studies of this multi-domain sensor and its mutants provide insights into signal regulation of second-messenger levels.
- Department of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX, 75390, USA.
Organizational Affiliation: