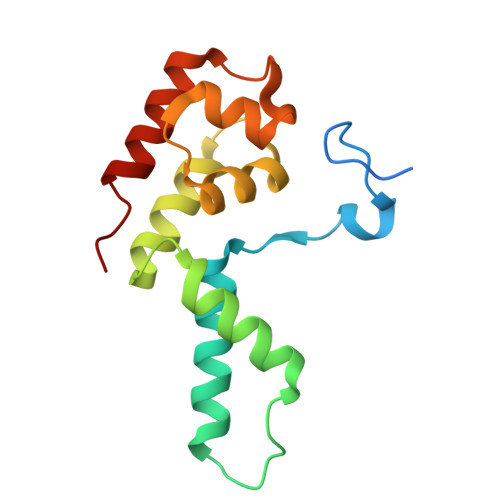
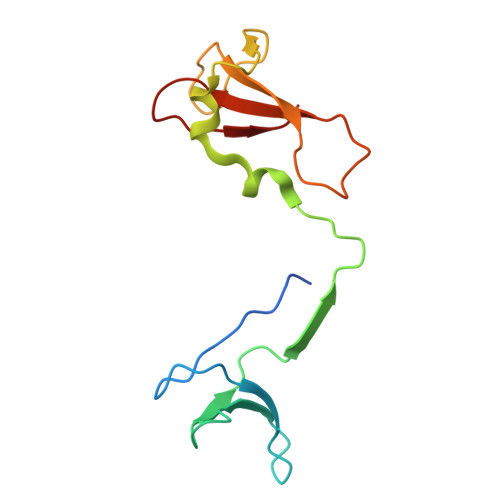
STK19 positions TFIIH for cell-free transcription-coupled DNA repair.
Mevissen, T.E.T., Kummecke, M., Schmid, E.W., Farnung, L., Walter, J.C.(2024) Cell 187: 7091-7106.e24
- PubMed: 39547228 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2024.10.020
- Primary Citation Related Structures:
9BZ0 - PubMed Abstract:
In transcription-coupled nucleotide excision repair (TC-NER), stalled RNA polymerase II (RNA Pol II) binds CSB and CRL4 CSA , which cooperate with UVSSA and ELOF1 to recruit TFIIH. To explore the mechanism of TC-NER, we recapitulated this reaction in vitro. When a plasmid containing a site-specific lesion is transcribed in frog egg extract, error-free repair is observed that depends on CSB, CRL4 CSA , UVSSA, and ELOF1. Repair also requires STK19, a factor previously implicated in transcription recovery after UV exposure. A 1.9-Å cryo-electron microscopy structure shows that STK19 binds the TC-NER complex through CSA and the RPB1 subunit of RNA Pol II. Furthermore, AlphaFold predicts that STK19 interacts with the XPD subunit of TFIIH, and disrupting this interface impairs cell-free repair. Molecular modeling suggests that STK19 positions TFIIH ahead of RNA Pol II for lesion verification. Our analysis of cell-free TC-NER suggests that STK19 couples RNA Pol II stalling to downstream repair events.
- Department of Biological Chemistry and Molecular Pharmacology, Blavatnik Institute, Harvard Medical School, Boston, MA 02115, USA; Howard Hughes Medical Institute, Boston, MA, USA.
Organizational Affiliation: