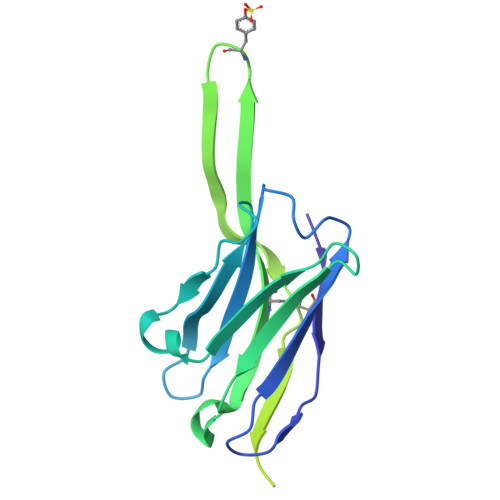
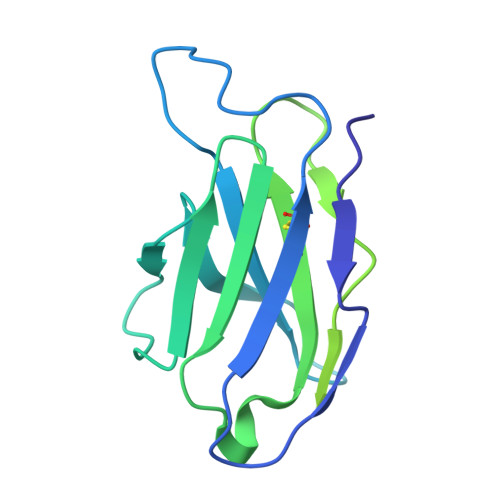
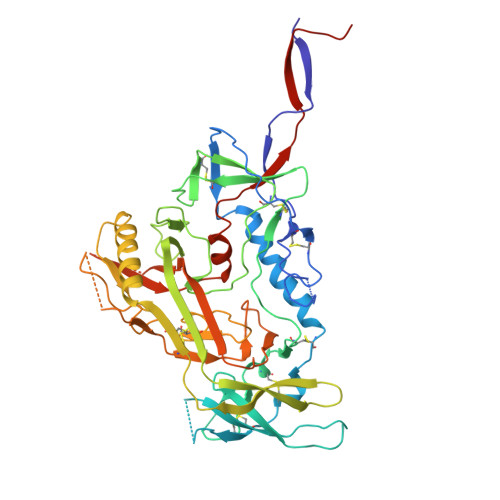
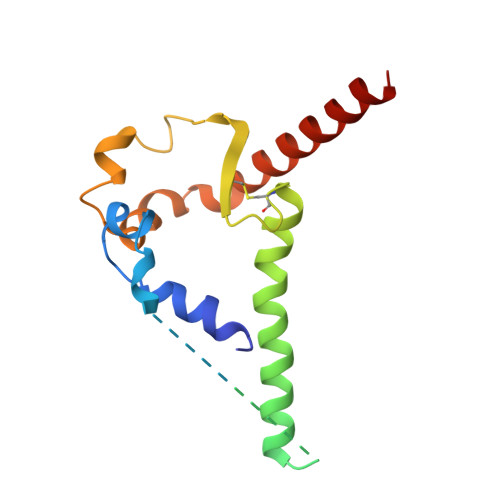
Structural and genetic basis of HIV-1 envelope V2 apex recognition by rhesus broadly neutralizing antibodies.
Roark, R.S., Habib, R., Gorman, J., Li, H., Connell, A.J., Bonsignori, M., Guo, Y., Hogarty, M.P., Olia, A.S., Sowers, K.J., Zhang, B., Bibollet-Ruche, F., Bylund, T., Callaghan, S., Carey, J.W., Cerutti, G., Harris, D.R., He, W., Lewis, E., Liu, T., Mason, R.D., Qiao, Y., Park, Y., Rando, J.M., Singh, A., Wolff, J.J., Lei, Q.P., Louder, M.K., Andrabi, R., Doria-Rose, N.A., Saunders, K.O., Seaman, M.S., Haynes, B.F., Kulp, D.W., Mascola, J.R., Roederer, M., Pierson, T.C., Sheng, Z., Hahn, B.H., Shaw, G.M., Kwong, P.D., Shapiro, L.(2025) J Exp Medicine 222
- PubMed: 40824240 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1084/jem.20250638
- Primary Citation Related Structures:
9BNK, 9BNL, 9BNM, 9BNP, 9BTH, 9BTI, 9BTJ, 9BTL, 9BTV - PubMed Abstract:
Broadly neutralizing antibodies targeting the V2 apex of HIV-1 envelope are desired as vaccine design templates, but few have been described. Here, we report 11 lineages of V2 apex-neutralizing antibodies from simian-human immunodeficiency virus (SHIV)-infected rhesus macaques and determine cryo-EM structures for 9. A single V2 apex-neutralizing lineage accounted for cross-clade breadth in most macaques, and somatic hypermutation relative to breadth was generally low, exemplified by antibody V033-a.01 with <5% nucleotide mutation and 37% breadth (208-strain panel). Envelope complex structures revealed eight different antibody classes (one multi-donor) and the complete repertoire of all five possible recognition topologies, recapitulating canonical human modes of apex insertion and C-strand hydrogen bonding. Despite this diversity in recognition, all rhesus-V2 apex antibodies were derived from reading frame two of the DH3-15*01 gene. Collectively, these results define-in rhesus-the structural and genetic basis of HIV-1 V2 apex recognition and demonstrate unprecedented structural plasticity of a highly selected immunogenetic element.
- Aaron Diamond AIDS Research Center, Columbia University Vagelos College of Physicians and Surgeons , New York, NY, USA.
Organizational Affiliation: