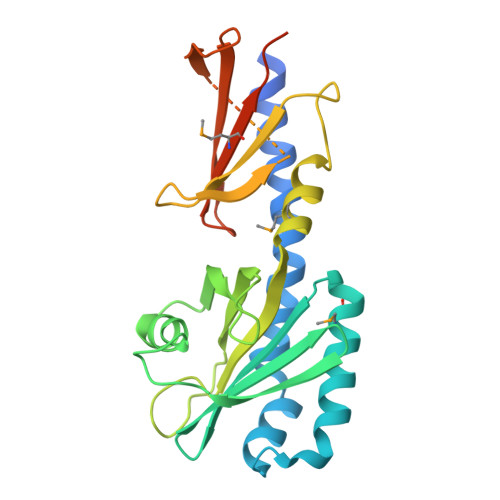
The chemoreceptor controlling the Wsp-like transduction pathway in Halomonas titanicae KHS3 binds and responds to purine derivatives.
Ramos Ricciuti, F.E., Soldano, A., Herrera Seitz, M.K., Gasperotti, A.F., Boyko, A., Jung, K., Bellinzoni, M., Lisa, M.N., Studdert, C.A.(2025) FEBS J 292: 1034-1051
- PubMed: 39529381 Search on PubMed
- DOI: https://doi.org/10.1111/febs.17320
- Primary Citation Related Structures:
9B9S, 9B9X, 9BA3 - PubMed Abstract:
The chemosensory pathway HtChe2 from the marine bacterium Halomonas titanicae KHS3 controls the activity of a diguanylate cyclase. Constitutive activation of this pathway results in colony morphology alterations and an increased ability to form biofilm. Such characteristics resemble the behavior of the Wsp pathway of Pseudomonas. In this work, we investigate the specificity of Htc10, the only chemoreceptor coded within the HtChe2 gene cluster. The purine derivatives guanine and hypoxanthine were identified as ligands of the recombinantly produced Htc10 ligand-binding domain, with dissociation constants in the micromolar range, and its structure was solved by X-ray protein crystallography. The sensor domain of Htc10 adopts a double Cache folding, with ligands bound to the membrane-distal pocket. A high-resolution structure of the occupied guanine-binding pocket allowed the identification of residues involved in ligand recognition. Such residues were validated by site-directed mutagenesis and isothermal titration calorimetry analyses of the protein variants. Moreover, heterologous expression of Htc10 in a Pseudomonas putida mutant lacking the native Wsp chemoreceptor promoted biofilm formation, a phenotype that was further enhanced by Htc10-specific ligands. To our knowledge, this is the first description of binding specificity of a chemoreceptor that controls the activity of an associated diguanylate cyclase, opening the way for dynamic studies of the signaling behavior of this kind of sensory complex.
- Instituto de Agrobiotecnología del Litoral (IAL, CONICET-UNL), Santa Fe, Argentina.
Organizational Affiliation: