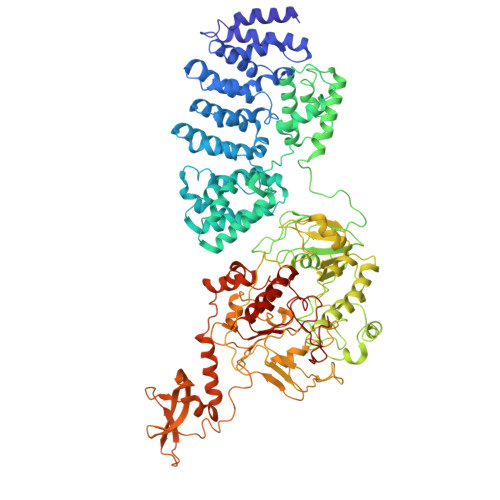
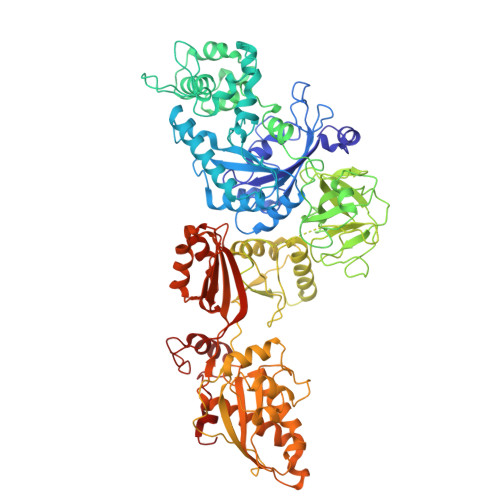
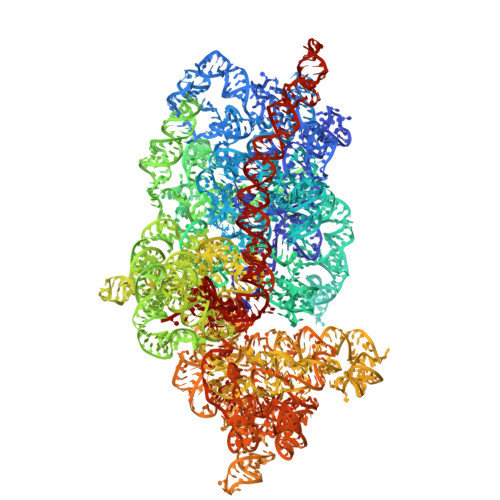
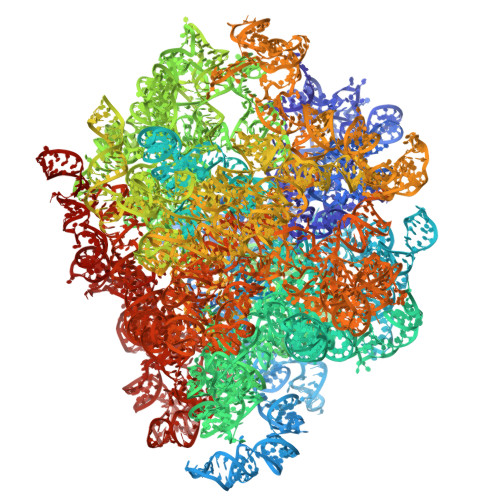
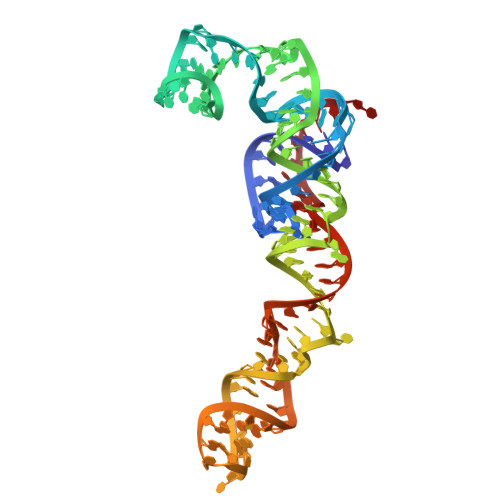
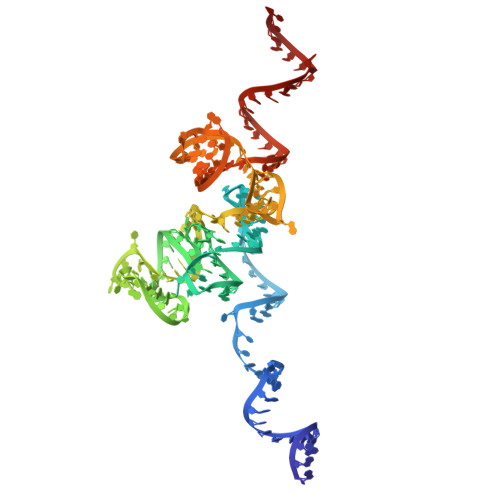
Capturing eukaryotic ribosome dynamics in situ at high resolution.
Cheng, J., Wu, C., Li, J., Yang, Q., Zhao, M., Zhang, X.(2025) Nat Struct Mol Biol 32: 698-708
- PubMed: 39789210 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-024-01454-9
- Primary Citation Related Structures:
8XU8, 8YLD, 8YLR, 8Z70, 8Z71 - PubMed Abstract:
Many protein complexes are highly dynamic in cells; thus, characterizing their conformational changes in cells is crucial for unraveling their functions. Here, using cryo-electron microscopy, 451,700 ribosome particles from Saccharomyces cerevisiae cell lamellae were obtained to solve the 60S region to 2.9-Å resolution by in situ single-particle analysis. Over 20 distinct conformations were identified by three-dimensional classification with resolutions typically higher than 4 Å. These conformations were used to reconstruct a complete elongation cycle of eukaryotic translation with elongation factors (eEFs). We found that compact eEF2 anchors to the partially rotated ribosome after subunit rolling and hypothesize that it stabilizes the local conformation for peptidyl transfer. Moreover, open-eEF3 binding to a fully rotated ribosome was observed, whose conformational change was coupled with head swiveling and body back-rotation of the 40S subunit.
- Key Laboratory of Biomacromolecules (CAS), National Laboratory of Biomacromolecules, CAS Center for Excellence in Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
Organizational Affiliation: