Structure and dynamics of cellodextrin phosphorylase from Clostridium thermocellum determine chain length and crystalline packing of highly ordered cellulose II synthesized in vitro
Kuga, T., Sunagawa, N., Igarashi, K.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cellodextrin phosphorylase | 990 | Acetivibrio thermocellus | Mutation(s): 0 Gene Names: cdp-ym4 | 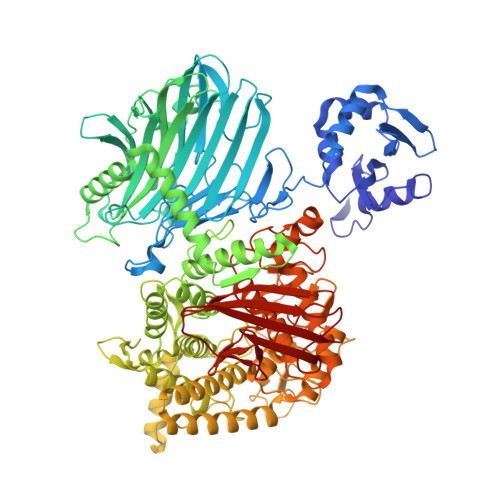 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q93HT8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| beta-D-glucopyranose-(1-4)-beta-D-glucopyranose-(1-4)-beta-D-glucopyranose-(1-4)-beta-D-glucopyranose-(1-4)-beta-D-glucopyranose | C [auth D] | 5 |  | N/A | |
Glycosylation Resources | |||||
GlyTouCan: G91683DU GlyCosmos: G91683DU | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CL (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A], F [auth A], G [auth B], H [auth B], I [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900016 Query on PRD_900016 | C [auth D] | beta-cellopentaose | Oligosaccharide / Metabolism |  |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900021 Query on PRD_900021 | D [auth C] | beta-cellotriose | Oligosaccharide / Metabolism |  |
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | cryoSPARC | 4.4.0 |
| MODEL REFINEMENT | PHENIX | 1.20 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Japan Society for the Promotion of Science (JSPS) | Japan | 22J12566 |
| Ministry of Education, Culture, Sports, Science and Technology (Japan) | Japan | 18H05494 |