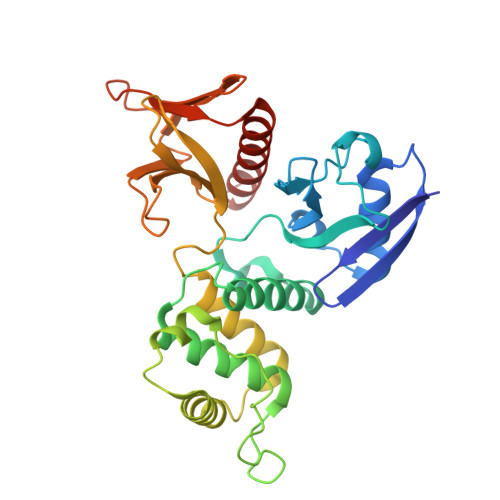
SARS-CoV-2 S assembly into virions facilitated by host ERM proteins.
Wang, J., Tai, W., Wang, Z., Dai, W., Yang, M., Guo, J., He, P., Nan, Y., Li, T., Zhou, S., Cui, D., Li, Y., Ma, C., Zhang, Y., Li, D., Zhu, Z., Chu, K., Wang, D., Yang, S., Zhuang, X., Tian, M., Huang, M., Zhang, X., Cheng, G., Ma, W.(2026) Proc Natl Acad Sci U S A 123: e2504517123-e2504517123
- PubMed: 41615754 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2504517123
- Primary Citation Related Structures:
8XZ0, 8XZ4, 8XZ5, 8XZ6 - PubMed Abstract:
The host cell cytoskeleton plays a critical role in the SARS-CoV-2 life cycle, though the underlying mechanisms remain poorly understood. This study investigates the interaction between the SARS-CoV-2 spike (S) protein and the cytoskeleton-associated ezrin-radixin-moesin (ERM) proteins through biochemical and structural characterization. A previously unidentified ERM-binding motif on the SARS-CoV-2 S protein is identified, revealing that S-ERM interactions are specifically conserved among highly pathogenic coronaviruses, including SARS-CoV, MERS-CoV, and SARS-CoV-2. Functionally, these interactions facilitate S packaging into virions by directing it to assembly sites, utilizing ERM's affinity for negatively curved membranes, akin to its role in cell surface protrusions. Silencing ERM expression significantly reduces SARS-CoV-2 titer, highlighting its essential role in viral propagation. Additionally, leveraging the established role of COPI-mediated trafficking in S localization, a compound is developed to disrupt S-COPI binding, promoting S secretion to the cell surface and effectively reducing viral titers. Our findings revealed a critical host-pathogen interaction that drives S incorporation into virions and identified ERM proteins as key facilitators of coronavirus assembly. Furthermore, our study suggests an antiviral strategy by targeting the S-COPI trafficking pathway. These insights advanced our understanding of coronavirus-host interactions and provided a potential therapeutic approach against SARS-CoV-2 and other highly pathogenic coronaviruses.
- School of Life Sciences, Beijing University of Chinese Medicine, Beijing 102488, China.
Organizational Affiliation: